更年期综合征(专业版)
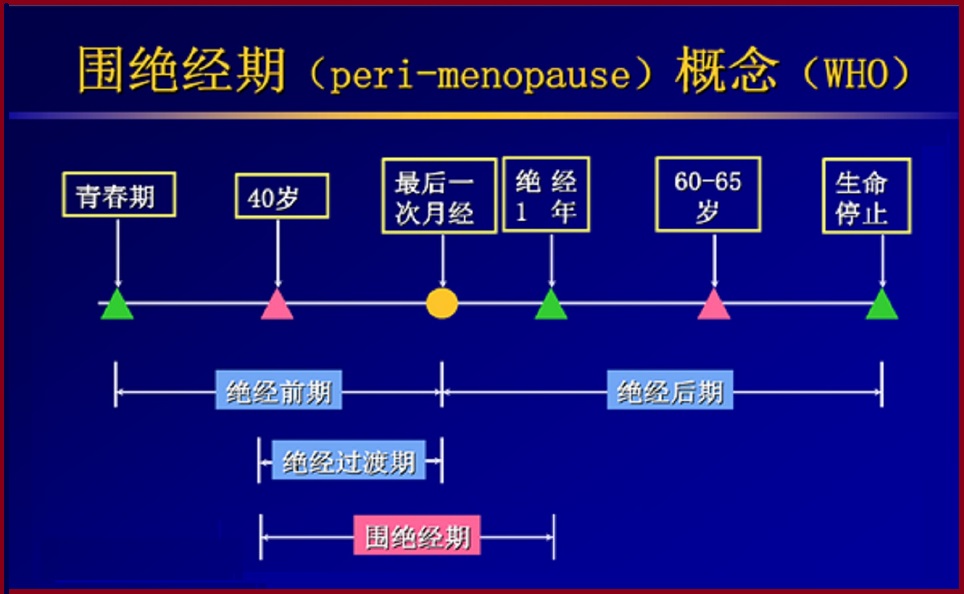
更年期(或称围绝经期)是月经永久结束的自然过程。月经开始发生变化,并持续几年后至连续停经一年,即确认为更年期结束(绝经)。
其他名称:围绝经期;更年期
英文名称:Menopause Syndrome,Perimenopause,Menopause
过早绝经的危险因素包括:
综合选项包括如下:
调整饮食与生活方式
营养与草本综合干预
以下是基于循证医学和循证营养学有关文献综合的结果。
有助于防控更年期综合征的营养和草本补充剂,主要包括如下:
1. 植物雌激素:
已知更年期的症状通常可以通过营养干预得到很好的控制。这些疗法中的许多都含有植物雌激素,这是一类弱激活雌激素受体的非甾体植物化合物,通常更喜欢ER-β而不是ER-α(详见下文“选择性雌激素受体调节”)1,2。
在生殖年龄期,当E2水平较高时,植物雌激素可能与E2竞争受体位点,从而具有抗雌激素作用;而在E2水平较低的更年期,植物雌激素通过刺激雌激素受体的能力(程度低于内源性雌激素)可能具有促雌激素作用3。换句话说,植物雌激素是雌激素受体的部分激动剂。一些植物雌激素还具有抗氧化、抗炎、抗增殖和表观遗传学作用,这可能有助于它们的健康促进作用4,5。一般来说,植物雌激素可以减轻更年期症状,包括潮热和泌尿生殖系统症状,并保护代谢、骨骼和心血管组织免受雌激素耗竭的负面影响,不增加(或可能降低)子宫内膜或乳腺癌症风险1,5,6。
附注:选择性雌激素受体调节
雌激素通过与细胞上的雌激素受体(ER)相互作用而发挥作用。已知两种经典的ER亚型(ER-α和ER-β),直接影响核基因表达并引发不同的细胞反应7。尽管ER-α和ER-β对卵巢发育和功能以及心血管保护都很重要,但ER-α是雌激素对代谢以及乳房、子宫和骨骼影响的更突出的介质,而ER-β更多地参与介导雌激素的神经系统和免疫作用,并通过ER-α在乳房和子宫中对抗促增殖信号8,9。另一种类型的雌激素反应受体,称为G蛋白偶联雌激素受体-1(GPER-1),通过激活细胞内的次级信使分子间接发挥作用7。
临床前证据表明,ER-β激活刺激抗炎过程,可能有助于保护更年期后的血管和神经健康,而不会促进乳腺或子宫内膜组织中的细胞增殖10-12。大多数具有雌激素样作用的植物化合物(包括大豆和红三叶草中的异黄酮)的一个优点是,因为它们比ER-α受体更强烈地激活ER-β受体1,2。GPER-1似乎在降低炎症免疫活性和介导雌激素对心血管和认知健康的积极影响方面也发挥着重要作用7,10,11。
1). 黑升麻:
黑升麻在治疗更年期症状和其他女性健康问题方面有着悠久的历史。在一项包括163名有更年期症状的妇女的对照研究中,与不治疗相比,服用黑升麻提取物,剂量为20mg,每天两次,持续三个月,改善了潮热、睡眠和易怒情绪13。一项临床证据综述得出结论,每天40mg的标准化黑升麻异丙醇提取物(Remifemin,莉芙敏)对减少更年期潮热和改善情绪是安全有效的14。另一项综述检查了35项临床试验,共43,759项参与者(包括13,000多名接受黑升麻提取物治疗者)发现,黑升麻提取物在治疗更年期症状方面的作用与低剂量透皮E2疗法相似,并且通过将其与圣约翰草(一种常用于治疗抑郁症的植物)相结合,其对情绪症状的益处得到了增强。分析中包括的六项随机对照试验中,有四项每天使用40mg的剂量,一项每天使用8mg,另一项每天服用64–128mg。特定提取物的成分因试验而异15。此外,黑升麻也可能对血管和骨骼健康产生积极影响16-18。
黑升麻通常与其他草药成分或营养素结合,可能会增加其有益效果。例如,一项在220名有症状的更年期妇女中进行的为期12周的试验发现,在缓解更年期症状,特别是情绪症状方面,黑升麻(每天13mg)和红景天(每天400mg)的组合比单独的黑升麻(每天13mg和1000mg的高剂量)或安慰剂更有效19。一项针对101名更年期参与者的随机安慰剂对照试验发现,520mg黑升麻、400mg圣洁莓、100mg大豆异黄酮和500mg月见草油的组合,每天服用一次,持续12周,可以改善潮热和出汗、睡眠问题、情绪和易怒,以及C反应蛋白(炎症标志物)、LDL胆固醇和甘油三酯的水平20。在另一项有170名参与者的随机安慰剂对照试验中,一种包括黑升麻、圣洁莓、大豆、牛蒡和野山药的组合,以550mg的剂量每天两次,持续八周,减少更年期盗汗(但不是潮热)的频率和强度21。一项针对50名围绝经期和绝经后妇女的为期三个月的随机对照试验报告称,黑升麻、圣洁莓、红三叶草、当归、西洋参和水飞蓟的组合可使潮热减少73%,盗汗减少69%;在试验结束时,47%接受草药组合治疗的女性没有潮热,而安慰剂组只有19%22。
一些对黑升麻的研究报告发现了植物雌激素化合物,但这些化合物并没有在所有提取物中一致发现。黑升麻的其他成分已被证明对血清素和其他神经递质受体起作用,这可能有助于黑升麻缓解更年期潮热、减少焦虑和提高认知功能18,23。黑升麻提取物中存在的具有减少氧化应激和抗炎作用的化合物也被认为有助于保存骨量18。多项临床试验表明,黑升麻的安全性表明,它对乳腺或子宫内膜组织没有致突变性作用,不会阻碍他莫昔芬(Soltamox)(一种选择性雌激素受体调节剂[SERM])对乳腺癌症患者的治疗效果,并且没有肝毒性;此外,一些证据表明黑升麻可以提高癌症患者的无病生存率23,24。
2). 西伯利亚大黄:
西伯利亚大黄根的标准化提取物(ERr 731 ®)已用于治疗更年期症状数十年。西伯利亚大黄的植物雌激素已被证明优先激活ER-β,对ER-α的作用明显较弱,表明其在乳腺和子宫内膜组织中可能是安全的25-27。
在一项随机安慰剂对照试验中,包括112名有症状的围绝经期妇女,在12周内每天服用一片4mg ERr 731的妇女中,根据44分更年期评定量表(MRS)评定的更年期症状得分下降了14.6分,而服用安慰剂的妇女仅下降了3分28。两项类似试验,每项包括109名围绝经期妇女发现,每天4mg ERr 731可减少潮热、焦虑症状和更年期症状总分,并在12周后比安慰剂更有效地改善整体健康和幸福感29,30。在上述两项试验中,研究人员报告了MRS评定量表11种症状中每一种症状的变化:与服用安慰剂的女性相比,在这些试验中服用ERr 731的女性在12周后MRS的所有11个项目都显著减少28,29。6在一项后续研究中,最初109名参与者中的80人在48周的观察期内服用了相同剂量的ERr 731,而51人继续治疗48周;参与者报告说,在两个随访阶段,更年期症状都有所减少31。非对照试验的结果进一步表明,ERr 731可以安全有效地改善围绝经期妇女的症状32,33。
一项安全性审查检查了1993年至2014年间使用ERr 731的女性报告的不良事件,在此期间,每天售出约1.4亿剂。审查发现,不良事件相对罕见;大多数是过敏反应或消化系统症状。仅报告了两起严重事件,包括一例癌症(未报告癌症病例);目前尚不清楚这些严重事件是否与西伯利亚胡麻的使用有关25。
3).大豆异黄酮:
大豆富含异黄酮的植物雌激素化合物。大豆异黄酮主要有染料木素(又称金曲异黄酮)和大豆黄素。在某些个体中,大豆黄素被特定的肠道细菌代谢为另一种植物雌激素—雌马酚(Equol,下文有介绍)。大豆黄素的雌激素效价约为E2的1/10000,而染料木素和雌马酚的雌激素效价接近E2的1/1000。异黄酮的另一个重要特征是,与ER-α(主要在乳腺和子宫组织中发现)相比,它们更喜欢ER-β(主要在骨骼、泌尿生殖系统和心血管系统中发现)34。53
有证据表明,大豆异黄酮可以减少更年期潮热34。一项荟萃分析评估了五项随机对照试验的结果,这些试验在围绝经期和绝经后妇女中使用雌马酚,或在被证实为雌马酚产生者的妇女中使用大豆异黄酮。分析发现,服用10-20mg雌马酚或高达200mg异黄酮(仅雌马酚产生者)的女性更年期潮热减少35。其他随机对照试验发现,每天服用54mg染料木素12个月,可以有效减少潮热,而不会引发子宫内膜增厚36,37。
大豆异黄酮对绝经后妇女具有其他有益作用,如减缓骨质流失38,改善脂质水平39和葡萄糖代谢38,降低心脏病风险40。尽管多项临床试验表明,膳食中摄入大量大豆异黄酮可降低患乳腺癌的总体风险41,但异黄酮补充剂对乳腺癌症风险的可能保护作用,可能仅限于雌激素受体表达(ER+)肿瘤以及处于围绝经期或绝经后早期的女性42。
附注:
雌马酚(Equol)是一种由大豆黄素(或大豆苷元)通过肠道细菌产生的异黄酮代谢产物43。62 Equol更稳定,更容易吸收,具有更高的生物利用度,以及比大豆黄素更强的植物雌激素活性43-45。它也是异黄酮衍生化合物中最有效的抗氧化剂44,45。人们认为产生雌马酚的能力可能与遗传因素和肠道微生物组构成的组合有关44。在亚洲人群中,50-70%的人是雌马酚产生者,而只有20-30%的西方人被发现生产Equol45。 Equol产生者在西方人群中的低流行率被认为是研究人员未能始终将食用大豆与心血管和其他健康益处联系起来的原因之一43,45。
4). 红三叶草:
红三叶草是一种生长在世界各地的常见植物,是异黄酮植物雌激素的来源,如鹰嘴豆素A、染料木素和大豆黄素。红三叶草已被研究其在治疗更年期症状方面的潜在益处,并已被证明具有抗炎和减少氧化应激的特性46。
一项对8项安慰剂对照试验的荟萃分析发现,红三叶草可以有效降低潮热的发生率;效果最好的试验包括每天经历5次或5次以上潮热的女性、每天使用80mg或以上红三叶草异黄酮的女性、报告鹰嘴豆素A浓度较高的女性,以及那些持续12周或更长时间的试验47。另一项荟萃分析包括三项使用标准化红三叶草异黄酮提取物(Promensil)的试验,剂量为每天80mg,发现其具有显著的减少潮热效果48。红三叶草还可以通过改善脂质状况和减少血管炎症来改善心血管健康49-51。
5). 啤酒花:
啤酒花可能因其对啤酒独特味道的贡献而闻名,但传统上也被用作镇静剂和治疗更年期症状。啤酒花的主要活性成分是烯酰化类黄酮(Prenylated flavonoids)化合物52,53。其中,8-异戊烯基柚皮素(8-prenylnaringenin)尤其表现出相对强大的植物雌激素活性,其与ER-α的相互作用比与ER-β的相互作用更强。另一方面,在实验室中,8-异戊烯基柚皮素也被发现可以抑制芳香化酶,这是一种参与雌激素合成的酶,这种作用可能会降低E2水平。实验室研究还表明,啤酒花提取物可以改善新陈代谢,促进正常细胞死亡,减少炎症信号,增强解毒能力,提高抗氧化能力52。尽管动物研究表明,啤酒花不会强烈刺激子宫内膜和乳腺组织的增殖,据报道,绝经后妇女使用啤酒花会导致子宫内膜增厚和出血,对于有乳腺癌病史或有乳腺癌高风险的妇女,其长期安全性尚不确定53-55。
在一项随机安慰剂对照试验中,120名围绝经期或绝经后早期的参与者每天服用500mg啤酒花,提供100mcg植物雌激素、持续12周,减少了潮热的次数和总体症状评分53,56。另一项有67名参与者的试验发现,标准化的啤酒花提取物每天提供100mcg的8-异戊烯基柚皮素,在6周后比安慰剂更能减少潮热,但啤酒花和安慰剂之间的差异在12周后消失57。一项针对36名女性的16周交叉试验显示,啤酒花(每天提供100mcg的8-异戊烯基柚皮素)和安慰剂在第一个8周阶段后减轻更年期症状方面没有任何差异。但是,在第一阶段服用安慰剂和第二阶段服用啤酒花的女性在第16周的症状缓解程度高于那些服用啤酒花和安慰剂的女性,这表明安慰剂效应很强,啤酒花可能会带来好处53,58。
啤酒花可以与其他营养成分结合使用以提高功效。在一项针对78名有中度至重度更年期症状的女性的随机安慰剂对照试验中,每天190mg啤酒花和大豆提取物联合治疗12周,使更年期症状评分降低20.16分,而安慰剂组降低14.80分,子宫内膜厚度或激素水平没有变化59。由啤酒花提取物与透明质酸(一种结缔组织成分)和维生素E联合制成的阴道凝胶,每晚2.5g,持续一周,然后每周两次,持续11周,在一项对100名绝经后妇女进行的非对照试验中,缓解阴道干燥并改善所有阴道症状60。
6). 胡芦巴:
胡芦巴籽传统上用于治疗一系列疾病,包括高胆固醇水平、高糖水平和消化问题,以及促进母乳分泌、缓解经前综合征(PMS)和减少更年期症状61。胡芦巴含有植物雌激素薯蓣皂苷元(Diosgenin),可能有助于心脏保护,并具有神经保护和免疫调节作用62,63。在一项包括48名围绝经期妇女的随机安慰剂对照试验中,每天两次250mg胡芦巴提取物,持续42天,减轻了症状,尤其是潮热、盗汗、抑郁和失眠,并通过增加E2、黄体酮和睾酮水平,降低FSH和性激素共轭球蛋白(一种结合雌激素和睾酮的蛋白质,使它们失效)的水平改善了激素平衡64。另一项随机对照试验,包括88名有中度至重度更年期症状的参与者,发现与安慰剂相比,每天1000mg胡芦巴提取物90天,可以改善潮热和其他症状,提高生活质量,并增加E2水平65。一项试验发现,115名参与者在12周内每天接受600mg胡芦巴提取物或安慰剂治疗,胡芦巴可以减少潮热、盗汗、社会心理、身体和性症状,以及总体更年期症状得分,而不影响E2水平66。
临床证据表明,含有胡芦巴的阴道制剂可能有助于治疗更年期相关的阴道症状。在一项试验中,60名患有阴道萎缩的绝经后妇女服用了含有5%胡芦巴提取物的阴道乳膏或安慰剂;8周后,使用胡芦巴乳膏的患者的萎缩得到改善,症状减轻67。在另一项有60名参与者的试验中,0.5g的含5%胡芦巴阴道乳膏每周两次,持续12周,可改善阴道萎缩及其症状,但不如低剂量共轭雌激素阴道乳膏有效68。
7). 甘草:
甘草根在中国和阿育吠陀草药传统中有着悠久的使用历史69。甘草提取物含有植物雌激素化合物和许多其他生物活性物质。甘草衍生的化合物在临床前研究中已被证明具有多种作用,包括抗炎、免疫调节、抗菌、抗溃疡、抗凝血、护肝以及促进骨骼生长69-72。实验室研究表明,甘草植物雌激素结合雌激素受体位点的亲和力不超过E2的1/1000,并且优先选择ER-β,而其他甘草化合物具有抗雌激素作用;总的效果似乎在很大程度上取决于细胞类型73。特别是,甘草的主要植物雌激素甘草素(Liquiritigenin,甘草苷元)对ER-β的结合亲和力比ER-α强74,75。在一项研究中,据报道,甘草素对ER-β的亲和力是其对ER-α亲和力的13倍73。
在一项包括90名更年期潮热女性的安慰剂对照试验中,每天三次服用330mg甘草提取物,持续八周,降低了潮热的频率和强度,直到停止治疗两周后效果才减弱76。一项由60名参与者参与的随机临床试验将每天1140mg甘草与标准激素替代疗法(HRT)进行了比较,其中每天服用HRT(0.312mg共轭雌激素加2.5mg甲孕酮),持续90天,用于治疗更年期潮热。甘草和HRT在减少潮热次数和持续时间方面同样有效,但HRT在降低潮热强度方面更有效77。在一项针对70名阴道萎缩女性的试验中,用含有2%甘草的阴道乳膏治疗8周,在改善阴道细胞健康和阴道萎缩症状方面比安慰剂更有效78。
值得注意的是,长期、高剂量使用甘草会导致钠和水的滞留以及钾的流失,从而可能导致血压升高和水肿79,80。负责这些负面作用的化合物被称为甘草酸。欧洲食品科学委员会建议,每日摄入的甘草甜酸应限制在100mg以下,相当于大约60-70g的粗甘草。尽管通常使用的甘草剂量对大多数人来说是安全的,但那些患有高血压、肾病或心脏病的人应该谨慎使用甘草,除非它是去甘草化的(通常称为“解甘草甜素DGL”)81。
8).当归:
当归是中医及东亚的传统草药,最常见的是与黄芪一起治疗更年期症状和其他女性生殖健康问题82,83。临床前证据表明,当归具有植物雌激素作用,与黄芪一起可促进骨骼生长84,85。
在一项针对100名有更年期症状的女性的随机安慰剂对照试验中,用传统配方的当归加黄芪治疗六个月,与安慰剂相比,可以明显减少轻度潮热,但更年期症状没有其他改善86。一项包括55名有症状的绝经后妇女的试验发现,当归加洋甘菊12周的联合服用,比安慰剂更能减少潮热、睡眠障碍和疲劳的次数和强度87。然而,在一项有71名参与者的研究中,24周后,与安慰剂相比,每天三次服用1.5g当归提取物对潮热或其他更年期症状没有影响88。
2. 碧萝芷:
法国海松皮提取物以碧萝芷的商品名广为人知,富含清除自由基的类黄酮原花青素。在一项对200名围绝经期参与者进行的随机安慰剂对照试验中,那些在六个月内每天服用200mg碧萝芷的参与者的所有更年期症状、血脂状况和抗氧化状态都有所改善89。一项对照试验包括38名有更年期症状的女性,每天服用100mg碧萝芷,以及33名连续8周没有服用的类似女性。该试验发现,服用碧萝芷的女性的六种常见症状(潮热、盗汗、情绪波动、睡眠困难、性欲低下、阴道干燥和月经失调)得分降低90。在一项随机安慰剂对照试验中,170名围绝经期妇女被分配接受30mg碧萝芷,每天两次或安慰剂治疗。12周后,碧萝芷组的更年期症状评分比安慰剂组的39%下降了56%,潮热和睡眠困难尤其得到改善91。
碧萝芷可能对健康有益,而不仅仅是缓解更年期症状。在35名绝经后妇女中,每天服用100mg碧萝芷治疗8周,不仅减轻了更年期症状,而且通过降低高血压以及胆固醇、甘油三酯、血糖、同型半胱氨酸、CRP和自由基水平,改善了心血管风险,而在35名对照组中没有发现这种变化92。一项针对43名绝经后骨质减少妇女的随机对照试验发现,12周后,与安慰剂相比,每天150mg不同的法国海松皮提取物改善了骨转换和抗氧化状态标志物93。碧萝芷加两种氨基酸(L-精氨酸和L-瓜氨酸)和玫瑰果提取物的组合。据报道,碧萝芷可改善围绝经期和绝经后妇女的阴道症状和性功能94。据报道,参与一项非对照试验的20名绝经后妇女在12周后也可改善皮肤弹性和水合作用95。
3. 圣洁莓:
圣洁莓被广泛用于治疗女性健康问题,包括月经失调、经前综合症、乳房疼痛、不孕不育和更年期症状96,97。尽管其浆果提取物最常使用,但也检查了圣洁莓叶精油对更年期症状的影响,据报道,它可能有益98,99。圣洁莓提取物已证明有能力激活神经系统中的多巴胺途径,从而抑制催乳素的释放,并有可能使激素周期正常化。此外,还发现圣洁莓可以在不改变正常昼夜节律的情况下增加褪黑激素的分泌,这可能有助于改善绝经后妇女的睡眠100,101。在一项针对52名有更年期症状参与者的随机安慰剂对照试验中,30mg圣洁莓提取物每天两次,持续8周,可以减少潮热、焦虑和更年期症状总分102。
圣洁莓常用于草药组合治疗更年期症状。例如,临床试验(如上所述)测试了圣洁莓与黑升麻、大豆异黄酮和月见草油,或黑升麻、大豆异黄酮、牛蒡和野山药的组合对更年期妇女的有益作用20,21。在一项开放的非对照试验中,在71名健康的绝经后妇女中,圣洁莓加大豆异黄酮和厚朴的组合使用12个月可以减轻潮热、情绪和睡眠症状。此外,治疗降低了血压、心率、同型半胱氨酸水平和血糖水平,并改善了胰岛素抵抗和炎症的标志物103。另一项有180名参与者参加的临床试验比较了圣洁莓(40mg)、大豆异黄酮(60mg)、厚朴(50mg)、产孢乳杆菌(109个孢子)和维生素D(35 mcg或1400IU)与大豆异黄酮单独使用12个月,发现该组合配方在降低潮热频率和强度、改善睡眠和心理健康方面更有效;两种治疗都没有引起子宫内膜或乳腺组织的变化104。
圣洁莓还可以提高绝经后妇女抗抑郁治疗的有效性。在一项为期8周的随机安慰剂对照试验中,包括46名接受西酞普兰(赛来沙)治疗的更年期妇女,与安慰剂相比,在治疗中添加圣洁莓(相当于每天1000mg干的圣洁莓浆果)加上黑孜然(每天500mg磨碎种子),可显著改善潮热、身体和社会心理功能105。
4. 圣约翰草:
圣约翰草(贯叶金丝桃)是一种以治疗情绪障碍,特别是抑郁症而闻名的植物106。对绝经后妇女进行的随机对照试验发现,圣约翰草治疗可降低潮热的强度和严重程度,并减轻抑郁症状的严重程度107-109。在一项试验中,47名有症状的围绝经期妇女在12周内每天三次服用900mg圣约翰草提取物,与安慰剂组相比,她们的生活质量更好,睡眠问题更少110。
圣约翰草和其他草药的组合也被研究对更年期症状的影响。在一项包括100名围绝经期和绝经后妇女的随机安慰剂对照试验中,圣约翰草(每天900mg)加圣洁莓提取物(每天1000mg)治疗16周对减少更年期症状无效111;然而,在一个由14名患有经前综合症症状的围绝经期晚期妇女组成的亚组中,这种草药组合减少了这些症状112。在一项观察性研究中,6141名更年期妇女服用圣约翰草加黑升麻或单独服用黑升麻6个月,圣约翰草和黑升麻与情绪症状的改善有关113。
5. 月见草油:
月见草油以其抗炎γ-亚油酸(GLA)含量相对较高而闻名,通常被推荐用于女性健康问题,如经前综合征、乳房疼痛、妊娠期糖尿病和更年期症状等114。在一项为期四周的随机安慰剂对照试验中,对100名有更年期症状的女性进行了研究,每天两次服用1000 mg月见草油(标准化为提供70-140mg GLA)的女性的心理症状评分下降了73%,但安慰剂组的心理症状得分没有变化115。
一项为期8周的试验中,189名女性每天服用1000mg月见草油或安慰剂,结果显示,服用月见草油的妇女与更年期有关的心理症状有所改善116。在一项有56名参与者的随机对照试验中,连续六周每天两次服用500 mg月见草油可降低更年期潮热的严重程度,但不会降低频率或持续时间,比安慰剂更有效117。一项由163名绝经后妇女完成的试验发现,与安慰剂相比,每天两次、每次1000mg月见草油在8周内可以降低盗汗的频率和严重程度118。此外,一项由35名妇女完成的小型试验发现,在服用40mg天然维生素E的同时,每天两次服用2000mg月见草油,持续6个月,对缓解潮热的效果并不比安慰剂好119。
6. 缬草:
已知缬草是一种以有益睡眠而闻名的植物。在一项针对60名更年期症状女性的随机安慰剂对照试验中,530mg缬草提取物每天两次,持续两个月,可降低潮热频率和严重程度120。在另一项针对68名更年期潮热女性的试验中,255mg缬草提取物,每天三次,持续八周,在降低潮热频率和强度方面比安慰剂更有效121。一项包括100名失眠的绝经后妇女的试验发现,530mg缬草提取物,每天两次,持续四周,比安慰剂更好地改善睡眠质量122。
7. 玛卡:
玛卡在南美洲历来用于治疗不孕不育和其他女性的荷尔蒙健康问题123。尽管临床研究有限,但一个研究小组进行了几项随机对照试验,发现玛卡对围绝经期和绝经后妇女可产生有益作用。在他们的第一项试点试验中,20名绝经后早期妇女每天两次服用1g糊化马卡粉片或安慰剂,为期两个月,8名受试者服用玛卡或安慰剂,持续八个月。与安慰剂相比,接受玛卡治疗的患者E2、孕酮和LH增加,FSH水平下降,与更年期相关的压力和不适减少;然而,研究人员确实注意到了强大的安慰剂效应124。同样的研究人员对20名围绝经期女性进行了为期四个月的交叉试验(每个参与者接受两个月的玛卡,每天两次,每次1g)和两个月安慰剂,按随机顺序),发现了更年期症状,如潮热、盗汗、睡眠问题、紧张、抑郁和心悸,以及改善的代谢参数在玛咖治疗期间减少了125。
在一项随机安慰剂对照试验中,124名绝经后早期的参与者每天两次服用1g糊化玛卡,持续三到四个月,增加了E2,降低了FSH水平,并减轻了更年期症状,尤其是潮热和盗汗126。一项涉及其中12名参与者的后续研究发现,使用玛卡与骨密度标志物水平的增加以及潮热、盗汗和应激反应标志物症状的改善有关127。在另一个研究小组进行的交叉试验中,在14名绝经后妇女中,与安慰剂相比,每天服用3.5g粉末状玛卡治疗6周后,心理和情绪症状以及性功能测试得分比安慰剂有所改善128。在另一项为期12周的交叉研究中,29名绝经后妇女被随机分为每天服用3.3g玛卡或安慰剂,每组服用6周,结果发现,与安慰剂相比,玛卡能减轻抑郁症状129。
8. 蜂王浆:
蜂王浆是一种营养丰富的物质,由保育蜂生产并喂食给蜂王一生和早期幼虫阶段130。与用作甜味剂的蜂蜜不同,蜂王浆富含蛋白质,通常不会被人类作为食物食用131。许多研究表明,蜂王浆具有抗炎、自由基猝灭、抗菌和免疫调节作用,并可能促进心血管、代谢和神经系统健康。此外,已经发现蜂王浆通过影响雌激素受体功能和引发表观遗传变化来影响女性生殖激素的活性,这对绝经期和绝经后妇女具有潜在的治疗作用131,132。
在一项针对200名绝经后妇女的随机对照试验中,每天服用1g蜂王浆,持续8周,比安慰剂更能降低更年期症状评分133。另一项针对42名绝经后女性的安慰剂对照试验发现,每天服用800mg酶处理的蜂王浆,连续12周,可显著减轻焦虑和背痛134。一项包括90名有泌尿生殖系统症状的绝经后妇女的试验将使用15%蜂王浆阴道膏与阴道雌激素疗法(结合马雌激素)或润滑剂的治疗进行了比较。三个月后,接受蜂王浆治疗的患者在性功能、泌尿功能和生活质量方面比接受雌激素或润滑剂治疗的患者有更大的改善,尽管实验室测试表明雌激素治疗对逆转阴道萎缩最有效135。
9.鼠尾草:
鼠尾草是一种常见的烹饪草本植物,传统上用于治疗女性激素相关的健康问题。研究发现,鼠尾草可以调节神经递质信号传导;对动物的研究表明,鼠尾草具有植物雌激素作用136-140。它还具有抗炎、止痛、清除自由基、抗菌、增强记忆以及降低血糖和胆固醇的作用141。
在一项对30名有症状的绝经后妇女进行的非对照试验中,连续四周每天服用100mg鼠尾草提取物,可以减轻潮热、盗汗、恐慌和疲劳的严重程度,并提高注意力142。在另一项非控制试验中,69名每天至少出现5次潮热的绝经后女性接受了280mg鼠尾草提取物的治疗,每天一次。八周后,她们的潮热频率和强度下降,身体功能、心理功能和泌尿生殖系统症状测试得分提高143。
更多内容可可点击其个性化综合干预方案如下:
更年期综合征管理:
更年期调理:
参阅本网如下专文了解更多相关内容:
医疗干预
一般治疗措施包括如下:
参考文献:
1. Rowe IJ et al. The effects of phytoestrogens on postmenopausal health. Climacteric : the journal of the International Menopause Society. Feb 2021;24(1):57-63.
2. Wang X et al. Exploring the Biological Activity and Mechanism of Xenoestrogens and Phytoestrogens in Cancers: Emerging Methods and Concepts. Int J Mol Sci. Aug 16 2021;22(16)
3. Farkas S et al. Estradiol and Estrogen-like Alternative Therapies in Use: The Importance of the Selective and Non-Classical Actions. Biomedicines. Apr 6 2022;10(4)
4. Petrine JCP, Del Bianco-Borges B. The influence of phytoestrogens on different physiological and pathological processes: An overview. Phytother Res. Jan 2021;35(1):180-197. doi:10.1002/ptr.6816. https://www.ncbi.nlm.nih.gov/pubmed/32780464
5. Domínguez-López I et al. Effects of Dietary Phytoestrogens on Hormones throughout a Human Lifespan: A Review. Nutrients. Aug 15 2020;12(8)doi:10.3390/nu12082456.
6. Abdi F et al. Impact of phytoestrogens on treatment of urogenital menopause symptoms: A systematic review of randomized clinical trials. European journal of obstetrics, gynecology, and reproductive biology. Jun 2021;261:222-235.
7. Dama A et al. Estrogen Receptor Functions and Pathways at the Vascular Immune Interface. Int J Mol Sci. Apr 20 2021;22(8)
8. Paterni I et al. Estrogen receptors alpha (ERalpha) and beta (ERbeta): subtype-selective ligands and clinical potential. Steroids. Nov 2014;90:13-29.
9. Jia M et al. Estrogen receptor alpha and beta in health and disease. Best Pract Res Clin Endocrinol Metab. Aug 2015;29(4):557-68.
10. McCarthy M et al. The peri-menopause in a woman's life: a systemic inflammatory phase that enables later neurodegenerative disease. J Neuroinflammation. Oct 23 2020;17(1):317.
11. da Silva JS et al. Estrogen Receptors: Therapeutic Perspectives for the Treatment of Cardiac Dysfunction after Myocardial Infarction. Int J Mol Sci. Jan 7 2021;22(2)
12. Vargas KG et al. The functions of estrogen receptor beta in the female brain: A systematic review. Maturitas. Nov 2016;93:41-57.
13. Guida M et al. Cimicifuga racemosa isopropanolic extract for menopausal symptoms: an observational prospective case-control study. Gynecological endocrinology. Dec 2021;37(12):1132-1137.
14. Castelo-Branco C et al. Black cohosh efficacy and safety for menopausal symptoms. The Spanish Menopause Society statement. Gynecological endocrinology. May 2022;38(5):379-384.
15. Castelo-Branco C et al. Review & meta-analysis: isopropanolic black cohosh extract iCR for menopausal symptoms - an update on the evidence. Climacteric. Apr 2021;24(2):109-119.
16. Fernandes ES et al. Effectiveness of the short-term use of Cimicifuga racemosa in the endothelial function of postmenopausal women: a double-blind, randomized, controlled trial. Climacteric. Jun 2020;23(3):245-251.
17. Gorach NV. Effects of cimicifuga racemosa on the hemodynamics parameters and quality of life in perimenopausal women with arterial hypertension. Wiadomosci lekarskie (Warsaw, Poland: 1960). 2018;71(5):1010-1014.
18. Mohapatra S et al. Benefits of Black Cohosh (Cimicifuga racemosa) for Women Health: An Up-Close and In-Depth Review. Pharmaceuticals (Basel). Feb 23 2022;15(3)
19. Pkhaladze L et al. Is More Effective in Combination with Rhodiola rosea L. for Relief of Menopausal Symptoms: A Randomized, Double-Blind, Placebo-Controlled Study. Pharmaceuticals (Basel). May 21 2020;13(5)
20. Rattanatantikul T et al. Efficacy and Safety of Nutraceutical on Menopausal Symptoms in Post-Menopausal Women: A Randomized, Double-Blind, Placebo-Controlled Clinical Trial. J Diet Suppl. 2022;19(2):168-183.
21. Shayan A et al. Effect of Combined Herbal Capsule Menohelp on Hot Flashes and Night Sweats in Postmenopausal Women: A Single-Blind Randomized Controlled Trial. Journal of menopausal medicine. Aug 2020;26(2):135-142.
22. Rotem C et al. Phyto-Female Complex for the relief of hot flushes, night sweats and quality of sleep: randomized, controlled, double-blind pilot study. Gynecological endocrinology. Feb 2007;23(2):117-22.
23. Henneicke-von Zepelin HH. 60 years of Cimicifuga racemosa medicinal products : Clinical research milestones, current study findings and current development. Wien Med Wochenschr. May 2017;167(7-8):147-159.
24. Ruan X et al. Benefit-risk profile of black cohosh (isopropanolic Cimicifuga racemosa extract) with and without St John's wort in breast cancer patients. Climacteric. Aug 2019;22(4):339-347.
25. Chang JL et al. Rheum rhaponticum Extract (ERr 731): Postmarketing Data on Safety Surveillance and Consumer Complaints. Integr Med (Encinitas). Jun 2016;15(3):34-9.
26. Wober J et al. Activation of estrogen receptor-beta by a special extract of Rheum rhaponticum (ERr 731), its aglycones and structurally related compounds. J Steroid Biochem Mol Biol. Nov-Dec 2007;107(3-5):191-201.
27. Moller F et al. Subtype-specific activation of estrogen receptors by a special extract of Rheum rhaponticum (ERr 731), its aglycones and structurally related compounds in U2OS human osteosarcoma cells. Phytomedicine. Nov 2007;14(11):716-26.
28. Kaszkin-Bettag M et al. Confirmation of the efficacy of ERr 731 in perimenopausal women with menopausal symptoms. Altern Ther Health Med. Jan-Feb 2009;15(1):24-34.
29. Heger M et al. Efficacy and safety of a special extract of Rheum rhaponticum (ERr 731) in perimenopausal women with climacteric complaints: a 12-week randomized, double-blind, placebo-controlled trial. Menopause. Sep-Oct 2006;13(5):744-59.
30. Kaszkin-Bettag M et al. The special extract ERr 731 of the roots of Rheum rhaponticum decreases anxiety and improves health state and general well-being in perimenopausal women. Menopause. Mar-Apr 2007;14(2):270-83.
31. Hasper I et al. Long-term efficacy and safety of the special extract ERr 731 of Rheum rhaponticum in perimenopausal women with menopausal symptoms. Menopause. Jan-Feb 2009;16(1):117-31.
32. Shah J et al. Evaluation of the Efficacy and Safety of Rheum rhaponticum Root Extract (ERr 731) for Menopausal Symptoms in Perimenopausal Indian Women: An Interim Analysis. Journal of mid-life health. Apr-Jun 2021;12(2):108-115.
33. Kaszkin-Bettag M et al. Efficacy of the special extract ERr 731 from rhapontic rhubarb for menopausal complaints: a 6-month open observational study. Altern Ther Health Med. Nov-Dec 2008;14(6):32-8.
34. Chen LR et al. Utilization of Isoflavones in Soybeans for Women with Menopausal Syndrome: An Overview. Int J Mol Sci. Mar 22 2021;22(6)
35. Daily JW et al. Equol Decreases Hot Flashes in Postmenopausal Women: A Systematic Review and Meta-Analysis of Randomized Clinical Trials. J Med Food. Feb 2019;22(2):127-139.
36. Crisafulli A et al. Effects of genistein on hot flushes in early postmenopausal women: a randomized, double-blind EPT- and placebo-controlled study. Menopause. Jul-Aug 2004;11(4):400-4.
37. D'Anna R et al. Effects of the phytoestrogen genistein on hot flushes, endometrium, and vaginal epithelium in postmenopausal women: a 2-year randomized, double-blind, placebo-controlled study. Menopause. Mar-Apr 2009;16(2):301-6.
38. Fang K et al. Soy isoflavones and glucose metabolism in menopausal women: A systematic review and meta-analysis of randomized controlled trials. Mol Nutr Food Res. Jul 2016;60(7):1602-14.
39. Baranska A et al. Effects of Soy Protein Containing of Isoflavones and Isoflavones Extract on Plasma Lipid Profile in Postmenopausal Women as a Potential Prevention Factor in Cardiovascular Diseases: Systematic Review and Meta-Analysis of Randomized Controlled Trials. Nutrients. Jul 24 2021;13(8)
40. Sathyapalan T et al. Soy isoflavones improve cardiovascular disease risk markers in women during the early menopause. Nutr Metab Cardiovasc Dis. Jul 2018;28(7):691-697.
41. Boutas I et al. Soy Isoflavones and Breast Cancer Risk: A Meta-analysis. In Vivo. Mar-Apr 2022;36(2):556-562.
42. Touillaud M et al. Use of dietary supplements containing soy isoflavones and breast cancer risk among women aged >50 y: a prospective study. Am J Clin Nutr. Mar 1 2019;109(3):597-605.
43. Leonard LM et al. Maximizing the Estrogenic Potential of Soy Isoflavones through the Gut Microbiome: Implication for Cardiometabolic Health in Postmenopausal Women. Nutrients. Jan 27 2022;14(3)
44. Mayo B et al. Equol: A Bacterial Metabolite from The Daidzein Isoflavone and Its Presumed Beneficial Health Effects. Nutrients. Sep 16 2019;11(9)
45. Sekikawa A et al. Effect of S-equol and Soy Isoflavones on Heart and Brain. Curr Cardiol Rev. 2019;15(2):114-135.
46. Mohsen A et al. Pharmacological and therapeutic properties of the Red Clover (Trifolium pratense L.): an overview of the new finding. J Tradit Chin Med. Aug 2021;41(4):642-649.
47. Kanadys W et al. Evaluation of Clinical Meaningfulness of Red Clover (Trifolium pratense L.) Extract to Relieve Hot Flushes and Menopausal Symptoms in Peri- and Post-Menopausal Women: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Nutrients. Apr 11 2021;13(4)
48. Myers SP et al. Effects of a standardised extract of Trifolium pratense (Promensil) at a dosage of 80mg in the treatment of menopausal hot flushes: A systematic review and meta-analysis. Phytomedicine. Jan 15 2017;24:141-147.
49. Blaszczuk A et al. Role of Phytoestrogen-Rich Bioactive Substances (Linum usitatissimum L., Glycine max L., Trifolium pratense L.) in Cardiovascular Disease Prevention in Postmenopausal Women: A Systematic Review and Meta-Analysis. Nutrients. Jun 14 2022;14(12)
50. Kanadys W et al. Effects of red clover (Trifolium pratense) isoflavones on the lipid profile of perimenopausal and postmenopausal women-A systematic review and meta-analysis. Maturitas. Feb 2020;132:7-16.
51. Wickham KA et al. Short-Term Supplementation With Fermented Red Clover Extract Reduces Vascular Inflammation in Early Post-menopausal Women. Front Cardiovasc Med. 2022;9:826959.
52. Bolton JL et al. The Multiple Biological Targets of Hops and Bioactive Compounds. Chemical research in toxicology. Feb 18 2019;32(2):222-233.
53. Stulikova K et al. Therapeutic Perspectives of 8-Prenylnaringenin, a Potent Phytoestrogen from Hops. Molecules. Mar 15 2018;23(3)
54. van Hunsel F et al Post-Menopausal Vaginal Hemorrhage Related to the Use of a Hop-Containing Phytotherapeutic Product. Drug Saf Case Rep. Dec 2015;2(1):14.
55. Keiler AM et al. Hop extracts and hop substances in treatment of menopausal complaints. Planta Med. May 2013;79(7):576-9.
56. Aghamiri V et al. The effect of Hop (Humulus lupulus L.) on early menopausal symptoms and hot flashes: A randomized placebo-controlled trial. Complement Ther Clin Pract. May 2016;23:130-5.
57. Heyerick A et al. A first prospective, randomized, double-blind, placebo-controlled study on the use of a standardized hop extract to alleviate menopausal discomforts. Maturitas. May 20 2006;54(2):164-75.
58. Erkkola R et al. A randomized, double-blind, placebo-controlled, cross-over pilot study on the use of a standardized hop extract to alleviate menopausal discomforts. Phytomedicine. May 2010;17(6):389-96.
59. Kim HI et al. Efficacy and Safety of a Standardized Soy and Hop Extract on Menopausal Symptoms: A 12-Week, Multicenter, Randomized, Double-Blind, Placebo-Controlled Clinical Trial. J Altern Complement Med. Nov 2021;27(11):959-967.
60. Morali G et al. Open, non-controlled clinical studies to assess the efficacy and safety of a medical device in form of gel topically and intravaginally used in postmenopausal women with genital atrophy. Arzneimittelforschung. 2006;56(3):230-8.
61. Sun W et al. Fenugreek Cultivation with Emphasis on Historical Aspects and its uses in Traditional Medicine and Modern Pharmaceutical Science. Mini Rev Med Chem. 2021;21(6):724-730.
62. Chen Y et al. Advances in the pharmacological activities and mechanisms of diosgenin. Chinese journal of natural medicines. Aug 2015;13(8):578-87.
63. Semwal P et al. Diosgenin: An Updated Pharmacological Review and Therapeutic Perspectives. Oxid Med Cell Longev. 2022;2022:1035441.
64. Khanna A et al. Efficacy of a novel extract of fenugreek seeds in alleviating vasomotor symptoms and depression in perimenopausal women: A randomized, double-blinded, placebo-controlled study. J Food Biochem. Dec 2020;44(12):e13507.
65. Shamshad Begum S et al. A Novel Extract of Fenugreek Husk (FenuSMART) Alleviates Postmenopausal Symptoms and Helps to Establish the Hormonal Balance: A Randomized, Double-Blind, Placebo-Controlled Study. Phytother Res. Nov 2016;30(11):1775-1784.
66. Steels E et al. Efficacy of a Proprietary Trigonella foenum-graecum L. De-Husked Seed Extract in Reducing Menopausal Symptoms in Otherwise Healthy Women: A Double-Blind, Randomized, Placebo-Controlled Study. Phytother Res. Sep 2017;31(9):1316-1322.
67. Mazalzadeh F et al. Effect of Trigonella foenum (fenugreek) vaginal cream on vaginal atrophy in postmenopausal women. J Family Med Prim Care. Jun 2020;9(6):2714-2719.
68. Safary M et al. Comparison of the Effects of Fenugreek Vaginal Cream and Ultra Low- Dose Estrogen on Atrophic Vaginitis. Curr Drug Deliv. 2020;17(9):815-822.
69. Wahab S et al. Glycyrrhiza glabra (Licorice): A Comprehensive Review on Its Phytochemistry, Biological Activities, Clinical Evidence and Toxicology. Plants (Basel). Dec 14 2021;10(12)
70. Minnetti M et al. Effects of licorice on sex hormones and the reproductive system. Nutrition. Nov-Dec 2022;103-104:111727.
71. Markina YV et al. Atheroprotective Effects of Glycyrrhiza glabra L. Molecules. Jul 22 2022;27(15)
72. Azizsoltani A et al. Ethyl Acetate Extract of Licorice Root (Glycyrrhiza glabra) Enhances Proliferation and Osteogenic Differentiation of Human Bone Marrow Mesenchymal Stem Cells. Iran J Pharm Res. Summer 2018;17(3):1057-1067.
73. Boonmuen N et al. Licorice root components in dietary supplements are selective estrogen receptor modulators with a spectrum of estrogenic and anti-estrogenic activities. Steroids. Jan 2016;105:42-9.
74. Hajirahimkhan A et al. Evaluation of estrogenic activity of licorice species in comparison with hops used in botanicals for menopausal symptoms. PLoS One. 2013;8(7):e67947.
75. Mersereau JE et al. Liquiritigenin is a plant-derived highly selective estrogen receptor beta agonist. Mol Cell Endocrinol. Feb 13 2008;283(1-2):49-57.
76. Nahidi F et al. Effects of licorice on relief and recurrence of menopausal hot flashes. Iran J Pharm Res. Spring 2012;11(2):541-8.
77. Menati L et al. Evaluation of contextual and demographic factors on licorice effects on reducing hot flashes in postmenopause women. Health Care Women Int. Jan 2014;35(1):87-99.
78. Sadeghi M et al. Impact of Glycyrrhiza glabra (licorice) vaginal cream on vaginal signs and symptoms of vaginal atrophy in postmenopausal women: A randomized double blind controlled trial. Journal of traditional and complementary medicine. Mar 2020;10(2):110-115.
79. Kwon YJ et al. A Review of the Pharmacological Efficacy and Safety of Licorice Root from Corroborative Clinical Trial Findings. J Med Food. Jan 2020;23(1):12-20.
80. McHugh J et al. A life-threatening case of pseudo-aldosteronism secondary to excessive liquorice ingestion. BMC endocrine disorders. Aug 6 2021;21(1):158.
81. Deutch MR et al. Bioactive Candy: Effects of Licorice on the Cardiovascular System. Foods. Oct 14 2019;8(10)
82. Lin HQ et al. Danggui Buxue Tang (Astragali Radix and Angelicae Sinensis Radix) for menopausal symptoms: A review. J Ethnopharmacol. Mar 6 2017;199:205-210.
83. Hook IL. Danggui to Angelica sinensis root: are potential benefits to European women lost in translation? A review. J Ethnopharmacol. Feb 27 2014;152(1):1-13.
84. Zhang WL et al. Chemical and biological assessment of Angelica herbal decoction: comparison of different preparations during historical applications. Phytomedicine. Aug 15 2012;19(11):1042-8.
85. Circosta C et al. Estrogenic activity of standardized extract of Angelica sinensis. Phytother Res. Aug 2006;20(8):665-9.
86. Haines CJ et al. A randomized, double-blind, placebo-controlled study of the effect of a Chinese herbal medicine preparation (Dang Gui Buxue Tang) on menopausal symptoms in Hong Kong Chinese women. Climacteric. Jun 2008;11(3):244-51.
87. Kupfersztain C et al. The immediate effect of natural plant extract, Angelica sinensis and Matricaria chamomilla (Climex) for the treatment of hot flushes during menopause. A preliminary report. Clinical and experimental obstetrics & gynecology. 2003;30(4):203-6.
88. Hirata JD et al. Does dong quai have estrogenic effects in postmenopausal women? A double-blind, placebo-controlled trial. Fertility and sterility. Dec 1997;68(6):981-6.
89. Yang HM et al. A randomised, double-blind, placebo-controlled trial on the effect of Pycnogenol on the climacteric syndrome in peri-menopausal women. Acta obstetricia et gynecologica Scandinavica. 2007;86(8):978-85.
90. Errichi S et al. Supplementation with Pycnogenol(R) improves signs and symptoms of menopausal transition. Panminerva Med. Sep 2011;53(3 Suppl 1):65-70.
91. Kohama T et al. Effect of low-dose French maritime pine bark extract on climacteric syndrome in 170 perimenopausal women: a randomized, double-blind, placebo-controlled trial. J Reprod Med. Jan-Feb 2013;58(1-2):39-46.
92. Luzzi R et al. Normalization of cardiovascular risk factors in peri-menopausal women with Pycnogenol(R). Minerva ginecologica. Feb 2017;69(1):29-34.
93. Majidi Z et al. Oligopin(R) Supplementation Mitigates Oxidative Stress in Postmenopausal Women with Osteopenia: A Randomized, Double-blind, Placebo-Controlled Trial. Phytomedicine. Jan 2021;81:153417.
94. Cesarone MR et al. Prevention of vaginal dryness in perimenopausal women. Supplementation with Lady Prelox(R). Minerva ginecologica. Dec 2019;71(6):434-441.
95. Marini A et al. Pycnogenol(R) effects on skin elasticity and hydration coincide with increased gene expressions of collagen type I and hyaluronic acid synthase in women. Skin Pharmacol Physiol. 2012;25(2):86-92.
96. Rani A et al. The genus Vitex: A review. Pharmacognosy reviews. Jul 2013;7(14):188-98.
97. Daniele C et al. Vitex agnus castus: a systematic review of adverse events. Drug safety. 2005;28(4):319-32.
98. Chopin Lucks B. Vitex agnus castus essential oil and menopausal balance: a research update [Complementary Therapies in Nursing and Midwifery 8 (2003) 148-154]. Complement Ther Nurs Midwifery. Aug 2003;9(3):157-60.
99. Lucks BC et al. Vitexagnus-castus essential oil and menopausal balance: a self-care survey. Complement Ther Nurs Midwifery. Aug 2002;8(3):148-54.
100. van Die MD et al. Vitex agnus-castus extracts for female reproductive disorders: a systematic review of clinical trials. Planta Med. May 2013;79(7):562-75.
101. Dericks-Tan JS et al. Dose-dependent stimulation of melatonin secretion after administration of Agnus castus. Exp Clin Endocrinol Diabetes. Feb 2003;111(1):44-6.
102. Naseri R et al. Comparison of Vitex agnus-castus Extracts with Placebo in Reducing Menopausal Symptoms: A Randomized Double-Blind Study. Korean J Fam Med. Nov 2019;40(6):362-367.
103. Maffei S et al. Long-term effects of a combination of isoflavones, agnus castus and magnolia extracts on climacteric symptoms and cardiometabolic risk profile in postmenopausal women. Gynecological endocrinology. Apr 2022;38(4):339-344.
104. De Franciscis P et al. Adding Agnus Castus and Magnolia to Soy Isoflavones Relieves Sleep Disturbances Besides Postmenopausal Vasomotor Symptoms-Long Term Safety and Effectiveness. Nutrients. Feb 13 2017;9(2)
105. Molaie M et al. Effects of a combination of Nigella sativa and Vitex agnus-castus with citalopram on healthy menopausal women with hot flashes: results from a subpopulation analysis. Gynecological endocrinology. Jan 2019;35(1):58-61.
106. Peterson B, Nguyen H. St. John's Wort. StatPearls. Publishing LLC.2023.
107. Eatemadnia A et al. The effect of Hypericum perforatum on postmenopausal symptoms and depression: A randomized controlled trial. Complement Ther Med. Aug 2019;45:109-113.
108. Abdali K et al. Effect of St John's wort on severity, frequency, and duration of hot flashes in premenopausal, perimenopausal and postmenopausal women: a randomized, double-blind, placebo-controlled study. Menopause. Mar 2010;17(2):326-31.
109. Liu YR et al. Hypericum perforatum L. preparations for menopause: a meta-analysis of efficacy and safety. Climacteric. Aug 2014;17(4):325-35.
110. Al-Akoum M et al. Effects of Hypericum perforatum (St. John's wort) on hot flashes and quality of life in perimenopausal women: a randomized pilot trial. Menopause. Mar-Apr 2009;16(2):307-14.
111. van Die MD et al. Hypericum perforatum with Vitex agnus-castus in menopausal symptoms: a randomized, controlled trial. Menopause. Jan-Feb 2009;16(1):156-63.
112. van Die MD et al. Effects of a combination of Hypericum perforatum and Vitex agnus-castus on PMS-like symptoms in late-perimenopausal women: findings from a subpopulation analysis. J Altern Complement Med. Sep 2009;15(9):1045-8.
113. Briese V et al. Black cohosh with or without St. John's wort for symptom-specific climacteric treatment--results of a large-scale, controlled, observational study. Maturitas. Aug 20 2007;57(4):405-14.
114. Mahboubi M. Evening Primrose (Oenothera biennis) Oil in Management of Female Ailments. Journal of menopausal medicine. Aug 2019;25(2):74-82.
115. Safdari F et al. Effect of Evening Primrose Oil on Postmenopausal Psychological Symptoms: A Triple-Blind Randomized Clinical Trial. Journal of menopausal medicine. Aug 2021;27(2):58-65.
116. Sharif SN et al. Impact of evening primrose oil consumption on psychological symptoms of postmenopausal women: a randomized double-blinded placebo-controlled clinical trial. Menopause. Feb 2020;27(2):194-198.
117. Farzaneh F et al. The effect of oral evening primrose oil on menopausal hot flashes: a randomized clinical trial. Archives of gynecology and obstetrics. Nov 2013;288(5):1075-9.
118. Kazemi F et al. The Effect of Evening Primrose Oil Capsule on Hot Flashes and Night Sweats in Postmenopausal Women: A Single-Blind Randomized Controlled Trial. Journal of menopausal medicine. Apr 2021;27(1):8-14.
119. Chenoy R et al. Effect of oral gamolenic acid from evening primrose oil on menopausal flushing. BMJ. Feb 19 1994;308(6927):501-3.
120. Jenabi E et al. The effect of Valerian on the severity and frequency of hot flashes: A triple-blind randomized clinical trial. Women Health. Mar 2018;58(3):297-304.
121. Mirabi P et al. The effects of valerian root on hot flashes in menopausal women. Iran J Pharm Res. Winter 2013;12(1):217-22.
122. Taavoni S et al. Effect of valerian on sleep quality in postmenopausal women: a randomized placebo-controlled clinical trial. Menopause. Sep 2011;18(9):951-5.
123. Lee MS et al. Maca (Lepidium meyenii) for treatment of menopausal symptoms: A systematic review. Maturitas. Nov 2011;70(3):227-33.
124. Meissner HO et al. Use of gelatinized maca (lepidium peruvianum) in early postmenopausal women. International journal of biomedical science. Jun 2005;1(1):33-45.
125. Meissner HO et al. Therapeutic Effects of Pre-Gelatinized Maca (Lepidium Peruvianum Chacon) used as a Non-Hormonal Alternative to HRT in Perimenopausal Women - Clinical Pilot Study. International journal of biomedical science. Jun 2006;2(2):143-59.
126. Meissner HO et al. Hormone-Balancing Effect of Pre-Gelatinized Organic Maca (Lepidium peruvianum Chacon): (II) Physiological and Symptomatic Responses of Early-Postmenopausal Women to Standardized doses of Maca in Double Blind, Randomized, Placebo-Controlled, Multi-Centre Clinical Study. International journal of biomedical science. Dec 2006;2(4):360-74.
127. Meissner HO et al. Hormone-Balancing Effect of Pre-Gelatinized Organic Maca (Lepidium peruvianum Chacon): (III) Clinical responses of early-postmenopausal women to Maca in double blind, randomized, Placebo-controlled, crossover configuration, outpatient study. International journal of biomedical science. Dec 2006;2(4):375-94.
128. Brooks NA et al. Beneficial effects of Lepidium meyenii (Maca) on psychological symptoms and measures of sexual dysfunction in postmenopausal women are not related to estrogen or androgen content. Menopause. Nov-Dec 2008;15(6):1157-62.
129. Stojanovska L et al. Maca reduces blood pressure and depression, in a pilot study in postmenopausal women. Climacteric. Feb 2015;18(1):69-78.
130. Ahmad S et al. New Insights into the Biological and Pharmaceutical Properties of Royal Jelly. Int J Mol Sci. Jan 8 2020;21(2)
131. Pasupuleti VR et al. Honey, Propolis, and Royal Jelly: A Comprehensive Review of Their Biological Actions and Health Benefits. Oxid Med Cell Longev. 2017;2017:1259510.
132. Balan A et al. Royal Jelly-A Traditional and Natural Remedy for Postmenopausal Symptoms and Aging-Related Pathologies. Molecules. Jul 20 2020;25(14)
133. Sharif SN et al. Effect of royal jelly on menopausal symptoms: A randomized placebo-controlled clinical trial. Complement Ther Clin Pract. Nov 2019;37:47-50.
134. Asama T et al. Royal Jelly Supplementation Improves Menopausal Symptoms Such as Backache, Low Back Pain, and Anxiety in Postmenopausal Japanese Women. Evid Based Complement Alternat Med. 2018;2018:4868412.
135. Seyyedi F et al. Comparison of the Effects of Vaginal Royal Jelly and Vaginal Estrogen on Quality of Life, Sexual and Urinary Function in Postmenopausal Women. J Clin Diagn Res. May 2016;10(5):QC01-5.
136. Tober C et al. Modulation of neurological pathways by Salvia officinalis and its dependence on manufacturing process and plant parts used. BMC Complement Altern Med. Jun 13 2019;19(1):128.
137. Margetts G et al. Evidence for anti-inflammatory effects and modulation of neurotransmitter metabolism by Salvia officinalis L. BMC Complement Med Ther. May 12 2022;22(1):131.
138. Elgayed SH et al. Estrogenic Effect of Salvia officinalis Extract on Reproductive Function of Female Mice and Identification of Its Phenolic Content. Combinatorial chemistry & high throughput screening. 2021;24(10):1654-1663.
139. Koubaa-Ghorbel F et al. Salvia officinalis mitigates uterus and liver damages induced by an estrogen deficiency in ovariectomized rats. J Food Biochem. May 2021;45(5):e13542.
140. Sabry MM et al. Estrogenic activity of Sage (Salvia officinalis L.) aerial parts and its isolated ferulic acid in immature ovariectomized female rats. J Ethnopharmacol. Jan 10 2022;282:114579.
141. Ghorbani A et al. Pharmacological properties of Salvia officinalis and its components. Journal of traditional and complementary medicine. Oct 2017;7(4):433-440.
142. Dadfar F et al. The effect of Saliva officinalis extract on the menopausal symptoms in postmenopausal women: An RCT. Int J Reprod Biomed. Apr 2019;17(4):287-92.
143. Bommer S et al. First time proof of sage's tolerability and efficacy in menopausal women with hot flushes. Adv Ther. Jun 2011;28(6):490-500.
美国妇女健康办公室
http://www.womenshealth.gov
美国梅奥诊所
www.mayoclinic.org
美国更年期学会
http://www.menopause.org
加拿大卫生部
http://www.hc-sc.gc.ca
加拿大女性健康网
http://www.womenshealthmatters.ca
其他参考文献:详见具体的综合干预方案
免责声明和安全信息
英文名称:Menopause Syndrome,Perimenopause,Menopause
定义
更年期(或称围绝经期)是月经永久结束的自然过程。月经开始发生变化,并持续几年后至连续停经一年,即确认为更年期结束(绝经)。绝经通常自然发生在52岁左右,但在40至60岁之间的任何时间都可以发生。40岁以前发生的更年期症状被认为更年期过早,又称为卵巢早衰。更年期是一个渐进的过程,它包括激素水平的变化和不规则的月经模式。病因
更年期综合征的主要原因是卵巢功能衰竭,卵巢分泌的雌激素逐渐减少,因而引发器官和组织的退行性变化,出现一系列的症状。月经由雌激素和黄体激素(孕酮)而发生,这些激素由卵巢释放。- 自然绝经是雌激素逐渐减少引起的,激素的减少最终会阻止卵巢释放卵子。
- 绝经也可以由手术去除子宫或卵巢引起,手术后绝经就立即出现了。
风险因素
更年期是与年龄衰老有关的生理自然发生过程。过早绝经的危险因素包括:
- 吸烟
- 环境污染
- 压力因素
- 放射或化疗
症状
更年期综合征可能包括如下:- 月经失调
- 潮热和盗汗
- 阴道干涩、性交疼痛
- 对性的兴趣降低
- 睡眠紊乱,如失眠
- 情绪变化,包括烦躁、焦虑或抑郁
- 皮肤干燥、脱发
- 尿失禁,尿频或漏尿
- 代谢缓慢,体重增加
并发症
更年期后,患某些疾病的风险增加包括:- 心血管疾病:主要由雌激素水平下降引起。
- 骨质酥松症:骨骼变脆和变弱,导致骨折风险增加,包括脊柱、臀部和手腕骨折等。
- 尿失禁与尿路感染:包括急迫性尿失禁、压力性尿失禁。
- 性生活障碍、性欲下降。
- 体重增加,新陈代谢减慢所致。
疗法
更年期是生命的一个自然组成部分,本身不需要治疗。性激素的快速下降,或雌、孕激素比失衡会导致更年期早期症状,一些治疗方法和生活方式的改变可能有助于减少引起问题的症状,并起到保护骨骼和心脏健康的作用。雌激素减少可增加心脏病和骨质疏松症的风险。综合选项包括如下:
调整饮食与生活方式
- 健康饮食,并富含膳食纤维如多种水果、蔬菜和全谷类食物等
- 限制饱和脂肪、反式脂肪、精加工食品和糖类
- 优质蛋白,如鸡、鱼、豆类或坚果
- 高钙食物,如牛奶、酸奶等
- 不要吸烟,或戒烟
- 户外活动,晒太阳可增加维生素D,防止骨松症
- 坚持运动、锻炼,有助于缓解压力
- 做好压力管理,缓解紧张焦虑,包括如呼吸练习、按摩、温水浴和安静的音乐等
- 维持健康体重
- 负重运动,包括步行、力量训练等,有助于保持骨密度、减少骨钙流失
- 咖啡因和酒精:可能会增加焦虑和失眠,并可能增加骨钙损失。如果喝酒,保持适量,即每天饮用1杯以下
- 辛辣的食物可能会使潮热更糟
- 吸烟会使绝经提前,引起心脏病和骨质疏松症风险。
营养与草本综合干预
以下是基于循证医学和循证营养学有关文献综合的结果。
有助于防控更年期综合征的营养和草本补充剂,主要包括如下:
1. 植物雌激素:
已知更年期的症状通常可以通过营养干预得到很好的控制。这些疗法中的许多都含有植物雌激素,这是一类弱激活雌激素受体的非甾体植物化合物,通常更喜欢ER-β而不是ER-α(详见下文“选择性雌激素受体调节”)1,2。
在生殖年龄期,当E2水平较高时,植物雌激素可能与E2竞争受体位点,从而具有抗雌激素作用;而在E2水平较低的更年期,植物雌激素通过刺激雌激素受体的能力(程度低于内源性雌激素)可能具有促雌激素作用3。换句话说,植物雌激素是雌激素受体的部分激动剂。一些植物雌激素还具有抗氧化、抗炎、抗增殖和表观遗传学作用,这可能有助于它们的健康促进作用4,5。一般来说,植物雌激素可以减轻更年期症状,包括潮热和泌尿生殖系统症状,并保护代谢、骨骼和心血管组织免受雌激素耗竭的负面影响,不增加(或可能降低)子宫内膜或乳腺癌症风险1,5,6。
附注:选择性雌激素受体调节
雌激素通过与细胞上的雌激素受体(ER)相互作用而发挥作用。已知两种经典的ER亚型(ER-α和ER-β),直接影响核基因表达并引发不同的细胞反应7。尽管ER-α和ER-β对卵巢发育和功能以及心血管保护都很重要,但ER-α是雌激素对代谢以及乳房、子宫和骨骼影响的更突出的介质,而ER-β更多地参与介导雌激素的神经系统和免疫作用,并通过ER-α在乳房和子宫中对抗促增殖信号8,9。另一种类型的雌激素反应受体,称为G蛋白偶联雌激素受体-1(GPER-1),通过激活细胞内的次级信使分子间接发挥作用7。
临床前证据表明,ER-β激活刺激抗炎过程,可能有助于保护更年期后的血管和神经健康,而不会促进乳腺或子宫内膜组织中的细胞增殖10-12。大多数具有雌激素样作用的植物化合物(包括大豆和红三叶草中的异黄酮)的一个优点是,因为它们比ER-α受体更强烈地激活ER-β受体1,2。GPER-1似乎在降低炎症免疫活性和介导雌激素对心血管和认知健康的积极影响方面也发挥着重要作用7,10,11。
1). 黑升麻:
黑升麻在治疗更年期症状和其他女性健康问题方面有着悠久的历史。在一项包括163名有更年期症状的妇女的对照研究中,与不治疗相比,服用黑升麻提取物,剂量为20mg,每天两次,持续三个月,改善了潮热、睡眠和易怒情绪13。一项临床证据综述得出结论,每天40mg的标准化黑升麻异丙醇提取物(Remifemin,莉芙敏)对减少更年期潮热和改善情绪是安全有效的14。另一项综述检查了35项临床试验,共43,759项参与者(包括13,000多名接受黑升麻提取物治疗者)发现,黑升麻提取物在治疗更年期症状方面的作用与低剂量透皮E2疗法相似,并且通过将其与圣约翰草(一种常用于治疗抑郁症的植物)相结合,其对情绪症状的益处得到了增强。分析中包括的六项随机对照试验中,有四项每天使用40mg的剂量,一项每天使用8mg,另一项每天服用64–128mg。特定提取物的成分因试验而异15。此外,黑升麻也可能对血管和骨骼健康产生积极影响16-18。
黑升麻通常与其他草药成分或营养素结合,可能会增加其有益效果。例如,一项在220名有症状的更年期妇女中进行的为期12周的试验发现,在缓解更年期症状,特别是情绪症状方面,黑升麻(每天13mg)和红景天(每天400mg)的组合比单独的黑升麻(每天13mg和1000mg的高剂量)或安慰剂更有效19。一项针对101名更年期参与者的随机安慰剂对照试验发现,520mg黑升麻、400mg圣洁莓、100mg大豆异黄酮和500mg月见草油的组合,每天服用一次,持续12周,可以改善潮热和出汗、睡眠问题、情绪和易怒,以及C反应蛋白(炎症标志物)、LDL胆固醇和甘油三酯的水平20。在另一项有170名参与者的随机安慰剂对照试验中,一种包括黑升麻、圣洁莓、大豆、牛蒡和野山药的组合,以550mg的剂量每天两次,持续八周,减少更年期盗汗(但不是潮热)的频率和强度21。一项针对50名围绝经期和绝经后妇女的为期三个月的随机对照试验报告称,黑升麻、圣洁莓、红三叶草、当归、西洋参和水飞蓟的组合可使潮热减少73%,盗汗减少69%;在试验结束时,47%接受草药组合治疗的女性没有潮热,而安慰剂组只有19%22。
一些对黑升麻的研究报告发现了植物雌激素化合物,但这些化合物并没有在所有提取物中一致发现。黑升麻的其他成分已被证明对血清素和其他神经递质受体起作用,这可能有助于黑升麻缓解更年期潮热、减少焦虑和提高认知功能18,23。黑升麻提取物中存在的具有减少氧化应激和抗炎作用的化合物也被认为有助于保存骨量18。多项临床试验表明,黑升麻的安全性表明,它对乳腺或子宫内膜组织没有致突变性作用,不会阻碍他莫昔芬(Soltamox)(一种选择性雌激素受体调节剂[SERM])对乳腺癌症患者的治疗效果,并且没有肝毒性;此外,一些证据表明黑升麻可以提高癌症患者的无病生存率23,24。
2). 西伯利亚大黄:
西伯利亚大黄根的标准化提取物(ERr 731 ®)已用于治疗更年期症状数十年。西伯利亚大黄的植物雌激素已被证明优先激活ER-β,对ER-α的作用明显较弱,表明其在乳腺和子宫内膜组织中可能是安全的25-27。
在一项随机安慰剂对照试验中,包括112名有症状的围绝经期妇女,在12周内每天服用一片4mg ERr 731的妇女中,根据44分更年期评定量表(MRS)评定的更年期症状得分下降了14.6分,而服用安慰剂的妇女仅下降了3分28。两项类似试验,每项包括109名围绝经期妇女发现,每天4mg ERr 731可减少潮热、焦虑症状和更年期症状总分,并在12周后比安慰剂更有效地改善整体健康和幸福感29,30。在上述两项试验中,研究人员报告了MRS评定量表11种症状中每一种症状的变化:与服用安慰剂的女性相比,在这些试验中服用ERr 731的女性在12周后MRS的所有11个项目都显著减少28,29。6在一项后续研究中,最初109名参与者中的80人在48周的观察期内服用了相同剂量的ERr 731,而51人继续治疗48周;参与者报告说,在两个随访阶段,更年期症状都有所减少31。非对照试验的结果进一步表明,ERr 731可以安全有效地改善围绝经期妇女的症状32,33。
一项安全性审查检查了1993年至2014年间使用ERr 731的女性报告的不良事件,在此期间,每天售出约1.4亿剂。审查发现,不良事件相对罕见;大多数是过敏反应或消化系统症状。仅报告了两起严重事件,包括一例癌症(未报告癌症病例);目前尚不清楚这些严重事件是否与西伯利亚胡麻的使用有关25。
3).大豆异黄酮:
大豆富含异黄酮的植物雌激素化合物。大豆异黄酮主要有染料木素(又称金曲异黄酮)和大豆黄素。在某些个体中,大豆黄素被特定的肠道细菌代谢为另一种植物雌激素—雌马酚(Equol,下文有介绍)。大豆黄素的雌激素效价约为E2的1/10000,而染料木素和雌马酚的雌激素效价接近E2的1/1000。异黄酮的另一个重要特征是,与ER-α(主要在乳腺和子宫组织中发现)相比,它们更喜欢ER-β(主要在骨骼、泌尿生殖系统和心血管系统中发现)34。53
有证据表明,大豆异黄酮可以减少更年期潮热34。一项荟萃分析评估了五项随机对照试验的结果,这些试验在围绝经期和绝经后妇女中使用雌马酚,或在被证实为雌马酚产生者的妇女中使用大豆异黄酮。分析发现,服用10-20mg雌马酚或高达200mg异黄酮(仅雌马酚产生者)的女性更年期潮热减少35。其他随机对照试验发现,每天服用54mg染料木素12个月,可以有效减少潮热,而不会引发子宫内膜增厚36,37。
大豆异黄酮对绝经后妇女具有其他有益作用,如减缓骨质流失38,改善脂质水平39和葡萄糖代谢38,降低心脏病风险40。尽管多项临床试验表明,膳食中摄入大量大豆异黄酮可降低患乳腺癌的总体风险41,但异黄酮补充剂对乳腺癌症风险的可能保护作用,可能仅限于雌激素受体表达(ER+)肿瘤以及处于围绝经期或绝经后早期的女性42。
附注:
雌马酚(Equol)是一种由大豆黄素(或大豆苷元)通过肠道细菌产生的异黄酮代谢产物43。62 Equol更稳定,更容易吸收,具有更高的生物利用度,以及比大豆黄素更强的植物雌激素活性43-45。它也是异黄酮衍生化合物中最有效的抗氧化剂44,45。人们认为产生雌马酚的能力可能与遗传因素和肠道微生物组构成的组合有关44。在亚洲人群中,50-70%的人是雌马酚产生者,而只有20-30%的西方人被发现生产Equol45。 Equol产生者在西方人群中的低流行率被认为是研究人员未能始终将食用大豆与心血管和其他健康益处联系起来的原因之一43,45。
4). 红三叶草:
红三叶草是一种生长在世界各地的常见植物,是异黄酮植物雌激素的来源,如鹰嘴豆素A、染料木素和大豆黄素。红三叶草已被研究其在治疗更年期症状方面的潜在益处,并已被证明具有抗炎和减少氧化应激的特性46。
一项对8项安慰剂对照试验的荟萃分析发现,红三叶草可以有效降低潮热的发生率;效果最好的试验包括每天经历5次或5次以上潮热的女性、每天使用80mg或以上红三叶草异黄酮的女性、报告鹰嘴豆素A浓度较高的女性,以及那些持续12周或更长时间的试验47。另一项荟萃分析包括三项使用标准化红三叶草异黄酮提取物(Promensil)的试验,剂量为每天80mg,发现其具有显著的减少潮热效果48。红三叶草还可以通过改善脂质状况和减少血管炎症来改善心血管健康49-51。
5). 啤酒花:
啤酒花可能因其对啤酒独特味道的贡献而闻名,但传统上也被用作镇静剂和治疗更年期症状。啤酒花的主要活性成分是烯酰化类黄酮(Prenylated flavonoids)化合物52,53。其中,8-异戊烯基柚皮素(8-prenylnaringenin)尤其表现出相对强大的植物雌激素活性,其与ER-α的相互作用比与ER-β的相互作用更强。另一方面,在实验室中,8-异戊烯基柚皮素也被发现可以抑制芳香化酶,这是一种参与雌激素合成的酶,这种作用可能会降低E2水平。实验室研究还表明,啤酒花提取物可以改善新陈代谢,促进正常细胞死亡,减少炎症信号,增强解毒能力,提高抗氧化能力52。尽管动物研究表明,啤酒花不会强烈刺激子宫内膜和乳腺组织的增殖,据报道,绝经后妇女使用啤酒花会导致子宫内膜增厚和出血,对于有乳腺癌病史或有乳腺癌高风险的妇女,其长期安全性尚不确定53-55。
在一项随机安慰剂对照试验中,120名围绝经期或绝经后早期的参与者每天服用500mg啤酒花,提供100mcg植物雌激素、持续12周,减少了潮热的次数和总体症状评分53,56。另一项有67名参与者的试验发现,标准化的啤酒花提取物每天提供100mcg的8-异戊烯基柚皮素,在6周后比安慰剂更能减少潮热,但啤酒花和安慰剂之间的差异在12周后消失57。一项针对36名女性的16周交叉试验显示,啤酒花(每天提供100mcg的8-异戊烯基柚皮素)和安慰剂在第一个8周阶段后减轻更年期症状方面没有任何差异。但是,在第一阶段服用安慰剂和第二阶段服用啤酒花的女性在第16周的症状缓解程度高于那些服用啤酒花和安慰剂的女性,这表明安慰剂效应很强,啤酒花可能会带来好处53,58。
啤酒花可以与其他营养成分结合使用以提高功效。在一项针对78名有中度至重度更年期症状的女性的随机安慰剂对照试验中,每天190mg啤酒花和大豆提取物联合治疗12周,使更年期症状评分降低20.16分,而安慰剂组降低14.80分,子宫内膜厚度或激素水平没有变化59。由啤酒花提取物与透明质酸(一种结缔组织成分)和维生素E联合制成的阴道凝胶,每晚2.5g,持续一周,然后每周两次,持续11周,在一项对100名绝经后妇女进行的非对照试验中,缓解阴道干燥并改善所有阴道症状60。
6). 胡芦巴:
胡芦巴籽传统上用于治疗一系列疾病,包括高胆固醇水平、高糖水平和消化问题,以及促进母乳分泌、缓解经前综合征(PMS)和减少更年期症状61。胡芦巴含有植物雌激素薯蓣皂苷元(Diosgenin),可能有助于心脏保护,并具有神经保护和免疫调节作用62,63。在一项包括48名围绝经期妇女的随机安慰剂对照试验中,每天两次250mg胡芦巴提取物,持续42天,减轻了症状,尤其是潮热、盗汗、抑郁和失眠,并通过增加E2、黄体酮和睾酮水平,降低FSH和性激素共轭球蛋白(一种结合雌激素和睾酮的蛋白质,使它们失效)的水平改善了激素平衡64。另一项随机对照试验,包括88名有中度至重度更年期症状的参与者,发现与安慰剂相比,每天1000mg胡芦巴提取物90天,可以改善潮热和其他症状,提高生活质量,并增加E2水平65。一项试验发现,115名参与者在12周内每天接受600mg胡芦巴提取物或安慰剂治疗,胡芦巴可以减少潮热、盗汗、社会心理、身体和性症状,以及总体更年期症状得分,而不影响E2水平66。
临床证据表明,含有胡芦巴的阴道制剂可能有助于治疗更年期相关的阴道症状。在一项试验中,60名患有阴道萎缩的绝经后妇女服用了含有5%胡芦巴提取物的阴道乳膏或安慰剂;8周后,使用胡芦巴乳膏的患者的萎缩得到改善,症状减轻67。在另一项有60名参与者的试验中,0.5g的含5%胡芦巴阴道乳膏每周两次,持续12周,可改善阴道萎缩及其症状,但不如低剂量共轭雌激素阴道乳膏有效68。
7). 甘草:
甘草根在中国和阿育吠陀草药传统中有着悠久的使用历史69。甘草提取物含有植物雌激素化合物和许多其他生物活性物质。甘草衍生的化合物在临床前研究中已被证明具有多种作用,包括抗炎、免疫调节、抗菌、抗溃疡、抗凝血、护肝以及促进骨骼生长69-72。实验室研究表明,甘草植物雌激素结合雌激素受体位点的亲和力不超过E2的1/1000,并且优先选择ER-β,而其他甘草化合物具有抗雌激素作用;总的效果似乎在很大程度上取决于细胞类型73。特别是,甘草的主要植物雌激素甘草素(Liquiritigenin,甘草苷元)对ER-β的结合亲和力比ER-α强74,75。在一项研究中,据报道,甘草素对ER-β的亲和力是其对ER-α亲和力的13倍73。
在一项包括90名更年期潮热女性的安慰剂对照试验中,每天三次服用330mg甘草提取物,持续八周,降低了潮热的频率和强度,直到停止治疗两周后效果才减弱76。一项由60名参与者参与的随机临床试验将每天1140mg甘草与标准激素替代疗法(HRT)进行了比较,其中每天服用HRT(0.312mg共轭雌激素加2.5mg甲孕酮),持续90天,用于治疗更年期潮热。甘草和HRT在减少潮热次数和持续时间方面同样有效,但HRT在降低潮热强度方面更有效77。在一项针对70名阴道萎缩女性的试验中,用含有2%甘草的阴道乳膏治疗8周,在改善阴道细胞健康和阴道萎缩症状方面比安慰剂更有效78。
值得注意的是,长期、高剂量使用甘草会导致钠和水的滞留以及钾的流失,从而可能导致血压升高和水肿79,80。负责这些负面作用的化合物被称为甘草酸。欧洲食品科学委员会建议,每日摄入的甘草甜酸应限制在100mg以下,相当于大约60-70g的粗甘草。尽管通常使用的甘草剂量对大多数人来说是安全的,但那些患有高血压、肾病或心脏病的人应该谨慎使用甘草,除非它是去甘草化的(通常称为“解甘草甜素DGL”)81。
8).当归:
当归是中医及东亚的传统草药,最常见的是与黄芪一起治疗更年期症状和其他女性生殖健康问题82,83。临床前证据表明,当归具有植物雌激素作用,与黄芪一起可促进骨骼生长84,85。
在一项针对100名有更年期症状的女性的随机安慰剂对照试验中,用传统配方的当归加黄芪治疗六个月,与安慰剂相比,可以明显减少轻度潮热,但更年期症状没有其他改善86。一项包括55名有症状的绝经后妇女的试验发现,当归加洋甘菊12周的联合服用,比安慰剂更能减少潮热、睡眠障碍和疲劳的次数和强度87。然而,在一项有71名参与者的研究中,24周后,与安慰剂相比,每天三次服用1.5g当归提取物对潮热或其他更年期症状没有影响88。
2. 碧萝芷:
法国海松皮提取物以碧萝芷的商品名广为人知,富含清除自由基的类黄酮原花青素。在一项对200名围绝经期参与者进行的随机安慰剂对照试验中,那些在六个月内每天服用200mg碧萝芷的参与者的所有更年期症状、血脂状况和抗氧化状态都有所改善89。一项对照试验包括38名有更年期症状的女性,每天服用100mg碧萝芷,以及33名连续8周没有服用的类似女性。该试验发现,服用碧萝芷的女性的六种常见症状(潮热、盗汗、情绪波动、睡眠困难、性欲低下、阴道干燥和月经失调)得分降低90。在一项随机安慰剂对照试验中,170名围绝经期妇女被分配接受30mg碧萝芷,每天两次或安慰剂治疗。12周后,碧萝芷组的更年期症状评分比安慰剂组的39%下降了56%,潮热和睡眠困难尤其得到改善91。
碧萝芷可能对健康有益,而不仅仅是缓解更年期症状。在35名绝经后妇女中,每天服用100mg碧萝芷治疗8周,不仅减轻了更年期症状,而且通过降低高血压以及胆固醇、甘油三酯、血糖、同型半胱氨酸、CRP和自由基水平,改善了心血管风险,而在35名对照组中没有发现这种变化92。一项针对43名绝经后骨质减少妇女的随机对照试验发现,12周后,与安慰剂相比,每天150mg不同的法国海松皮提取物改善了骨转换和抗氧化状态标志物93。碧萝芷加两种氨基酸(L-精氨酸和L-瓜氨酸)和玫瑰果提取物的组合。据报道,碧萝芷可改善围绝经期和绝经后妇女的阴道症状和性功能94。据报道,参与一项非对照试验的20名绝经后妇女在12周后也可改善皮肤弹性和水合作用95。
3. 圣洁莓:
圣洁莓被广泛用于治疗女性健康问题,包括月经失调、经前综合症、乳房疼痛、不孕不育和更年期症状96,97。尽管其浆果提取物最常使用,但也检查了圣洁莓叶精油对更年期症状的影响,据报道,它可能有益98,99。圣洁莓提取物已证明有能力激活神经系统中的多巴胺途径,从而抑制催乳素的释放,并有可能使激素周期正常化。此外,还发现圣洁莓可以在不改变正常昼夜节律的情况下增加褪黑激素的分泌,这可能有助于改善绝经后妇女的睡眠100,101。在一项针对52名有更年期症状参与者的随机安慰剂对照试验中,30mg圣洁莓提取物每天两次,持续8周,可以减少潮热、焦虑和更年期症状总分102。
圣洁莓常用于草药组合治疗更年期症状。例如,临床试验(如上所述)测试了圣洁莓与黑升麻、大豆异黄酮和月见草油,或黑升麻、大豆异黄酮、牛蒡和野山药的组合对更年期妇女的有益作用20,21。在一项开放的非对照试验中,在71名健康的绝经后妇女中,圣洁莓加大豆异黄酮和厚朴的组合使用12个月可以减轻潮热、情绪和睡眠症状。此外,治疗降低了血压、心率、同型半胱氨酸水平和血糖水平,并改善了胰岛素抵抗和炎症的标志物103。另一项有180名参与者参加的临床试验比较了圣洁莓(40mg)、大豆异黄酮(60mg)、厚朴(50mg)、产孢乳杆菌(109个孢子)和维生素D(35 mcg或1400IU)与大豆异黄酮单独使用12个月,发现该组合配方在降低潮热频率和强度、改善睡眠和心理健康方面更有效;两种治疗都没有引起子宫内膜或乳腺组织的变化104。
圣洁莓还可以提高绝经后妇女抗抑郁治疗的有效性。在一项为期8周的随机安慰剂对照试验中,包括46名接受西酞普兰(赛来沙)治疗的更年期妇女,与安慰剂相比,在治疗中添加圣洁莓(相当于每天1000mg干的圣洁莓浆果)加上黑孜然(每天500mg磨碎种子),可显著改善潮热、身体和社会心理功能105。
4. 圣约翰草:
圣约翰草(贯叶金丝桃)是一种以治疗情绪障碍,特别是抑郁症而闻名的植物106。对绝经后妇女进行的随机对照试验发现,圣约翰草治疗可降低潮热的强度和严重程度,并减轻抑郁症状的严重程度107-109。在一项试验中,47名有症状的围绝经期妇女在12周内每天三次服用900mg圣约翰草提取物,与安慰剂组相比,她们的生活质量更好,睡眠问题更少110。
圣约翰草和其他草药的组合也被研究对更年期症状的影响。在一项包括100名围绝经期和绝经后妇女的随机安慰剂对照试验中,圣约翰草(每天900mg)加圣洁莓提取物(每天1000mg)治疗16周对减少更年期症状无效111;然而,在一个由14名患有经前综合症症状的围绝经期晚期妇女组成的亚组中,这种草药组合减少了这些症状112。在一项观察性研究中,6141名更年期妇女服用圣约翰草加黑升麻或单独服用黑升麻6个月,圣约翰草和黑升麻与情绪症状的改善有关113。
5. 月见草油:
月见草油以其抗炎γ-亚油酸(GLA)含量相对较高而闻名,通常被推荐用于女性健康问题,如经前综合征、乳房疼痛、妊娠期糖尿病和更年期症状等114。在一项为期四周的随机安慰剂对照试验中,对100名有更年期症状的女性进行了研究,每天两次服用1000 mg月见草油(标准化为提供70-140mg GLA)的女性的心理症状评分下降了73%,但安慰剂组的心理症状得分没有变化115。
一项为期8周的试验中,189名女性每天服用1000mg月见草油或安慰剂,结果显示,服用月见草油的妇女与更年期有关的心理症状有所改善116。在一项有56名参与者的随机对照试验中,连续六周每天两次服用500 mg月见草油可降低更年期潮热的严重程度,但不会降低频率或持续时间,比安慰剂更有效117。一项由163名绝经后妇女完成的试验发现,与安慰剂相比,每天两次、每次1000mg月见草油在8周内可以降低盗汗的频率和严重程度118。此外,一项由35名妇女完成的小型试验发现,在服用40mg天然维生素E的同时,每天两次服用2000mg月见草油,持续6个月,对缓解潮热的效果并不比安慰剂好119。
6. 缬草:
已知缬草是一种以有益睡眠而闻名的植物。在一项针对60名更年期症状女性的随机安慰剂对照试验中,530mg缬草提取物每天两次,持续两个月,可降低潮热频率和严重程度120。在另一项针对68名更年期潮热女性的试验中,255mg缬草提取物,每天三次,持续八周,在降低潮热频率和强度方面比安慰剂更有效121。一项包括100名失眠的绝经后妇女的试验发现,530mg缬草提取物,每天两次,持续四周,比安慰剂更好地改善睡眠质量122。
7. 玛卡:
玛卡在南美洲历来用于治疗不孕不育和其他女性的荷尔蒙健康问题123。尽管临床研究有限,但一个研究小组进行了几项随机对照试验,发现玛卡对围绝经期和绝经后妇女可产生有益作用。在他们的第一项试点试验中,20名绝经后早期妇女每天两次服用1g糊化马卡粉片或安慰剂,为期两个月,8名受试者服用玛卡或安慰剂,持续八个月。与安慰剂相比,接受玛卡治疗的患者E2、孕酮和LH增加,FSH水平下降,与更年期相关的压力和不适减少;然而,研究人员确实注意到了强大的安慰剂效应124。同样的研究人员对20名围绝经期女性进行了为期四个月的交叉试验(每个参与者接受两个月的玛卡,每天两次,每次1g)和两个月安慰剂,按随机顺序),发现了更年期症状,如潮热、盗汗、睡眠问题、紧张、抑郁和心悸,以及改善的代谢参数在玛咖治疗期间减少了125。
在一项随机安慰剂对照试验中,124名绝经后早期的参与者每天两次服用1g糊化玛卡,持续三到四个月,增加了E2,降低了FSH水平,并减轻了更年期症状,尤其是潮热和盗汗126。一项涉及其中12名参与者的后续研究发现,使用玛卡与骨密度标志物水平的增加以及潮热、盗汗和应激反应标志物症状的改善有关127。在另一个研究小组进行的交叉试验中,在14名绝经后妇女中,与安慰剂相比,每天服用3.5g粉末状玛卡治疗6周后,心理和情绪症状以及性功能测试得分比安慰剂有所改善128。在另一项为期12周的交叉研究中,29名绝经后妇女被随机分为每天服用3.3g玛卡或安慰剂,每组服用6周,结果发现,与安慰剂相比,玛卡能减轻抑郁症状129。
8. 蜂王浆:
蜂王浆是一种营养丰富的物质,由保育蜂生产并喂食给蜂王一生和早期幼虫阶段130。与用作甜味剂的蜂蜜不同,蜂王浆富含蛋白质,通常不会被人类作为食物食用131。许多研究表明,蜂王浆具有抗炎、自由基猝灭、抗菌和免疫调节作用,并可能促进心血管、代谢和神经系统健康。此外,已经发现蜂王浆通过影响雌激素受体功能和引发表观遗传变化来影响女性生殖激素的活性,这对绝经期和绝经后妇女具有潜在的治疗作用131,132。
在一项针对200名绝经后妇女的随机对照试验中,每天服用1g蜂王浆,持续8周,比安慰剂更能降低更年期症状评分133。另一项针对42名绝经后女性的安慰剂对照试验发现,每天服用800mg酶处理的蜂王浆,连续12周,可显著减轻焦虑和背痛134。一项包括90名有泌尿生殖系统症状的绝经后妇女的试验将使用15%蜂王浆阴道膏与阴道雌激素疗法(结合马雌激素)或润滑剂的治疗进行了比较。三个月后,接受蜂王浆治疗的患者在性功能、泌尿功能和生活质量方面比接受雌激素或润滑剂治疗的患者有更大的改善,尽管实验室测试表明雌激素治疗对逆转阴道萎缩最有效135。
9.鼠尾草:
鼠尾草是一种常见的烹饪草本植物,传统上用于治疗女性激素相关的健康问题。研究发现,鼠尾草可以调节神经递质信号传导;对动物的研究表明,鼠尾草具有植物雌激素作用136-140。它还具有抗炎、止痛、清除自由基、抗菌、增强记忆以及降低血糖和胆固醇的作用141。
在一项对30名有症状的绝经后妇女进行的非对照试验中,连续四周每天服用100mg鼠尾草提取物,可以减轻潮热、盗汗、恐慌和疲劳的严重程度,并提高注意力142。在另一项非控制试验中,69名每天至少出现5次潮热的绝经后女性接受了280mg鼠尾草提取物的治疗,每天一次。八周后,她们的潮热频率和强度下降,身体功能、心理功能和泌尿生殖系统症状测试得分提高143。
更多内容可可点击其个性化综合干预方案如下:
更年期综合征管理:
更年期调理:
参阅本网如下专文了解更多相关内容:
医疗干预
一般治疗措施包括如下:
- 激素替代疗法(HRT):HRT药有助于平衡体内性激素、缓解症状,包括如下:
- 雌激素
- 孕酮
- 雌激素和孕激素组合
- 少量雄性激素
HRT药有片剂、凝胶、皮肤贴片、阴道环,注射和微丸插入皮肤等多种形式。
注意:激素可影响全身。虽然可以减少某些症状,但HRT也可能增加其他疾病的风险,包括如下:- 子宫癌
- 乳腺癌
- 卵巢癌
- 心脏病
- 血栓
- 药物治疗更年期症状如:
- 某些血压药物
- 抗癫痫药物
- 抗抑郁药物
- 阴道保湿剂和润滑剂,帮助减少阴道干涩。
预防
更年期是生命自然过程,不需要也不能预防。参考文献:
1. Rowe IJ et al. The effects of phytoestrogens on postmenopausal health. Climacteric : the journal of the International Menopause Society. Feb 2021;24(1):57-63.
2. Wang X et al. Exploring the Biological Activity and Mechanism of Xenoestrogens and Phytoestrogens in Cancers: Emerging Methods and Concepts. Int J Mol Sci. Aug 16 2021;22(16)
3. Farkas S et al. Estradiol and Estrogen-like Alternative Therapies in Use: The Importance of the Selective and Non-Classical Actions. Biomedicines. Apr 6 2022;10(4)
4. Petrine JCP, Del Bianco-Borges B. The influence of phytoestrogens on different physiological and pathological processes: An overview. Phytother Res. Jan 2021;35(1):180-197. doi:10.1002/ptr.6816. https://www.ncbi.nlm.nih.gov/pubmed/32780464
5. Domínguez-López I et al. Effects of Dietary Phytoestrogens on Hormones throughout a Human Lifespan: A Review. Nutrients. Aug 15 2020;12(8)doi:10.3390/nu12082456.
6. Abdi F et al. Impact of phytoestrogens on treatment of urogenital menopause symptoms: A systematic review of randomized clinical trials. European journal of obstetrics, gynecology, and reproductive biology. Jun 2021;261:222-235.
7. Dama A et al. Estrogen Receptor Functions and Pathways at the Vascular Immune Interface. Int J Mol Sci. Apr 20 2021;22(8)
8. Paterni I et al. Estrogen receptors alpha (ERalpha) and beta (ERbeta): subtype-selective ligands and clinical potential. Steroids. Nov 2014;90:13-29.
9. Jia M et al. Estrogen receptor alpha and beta in health and disease. Best Pract Res Clin Endocrinol Metab. Aug 2015;29(4):557-68.
10. McCarthy M et al. The peri-menopause in a woman's life: a systemic inflammatory phase that enables later neurodegenerative disease. J Neuroinflammation. Oct 23 2020;17(1):317.
11. da Silva JS et al. Estrogen Receptors: Therapeutic Perspectives for the Treatment of Cardiac Dysfunction after Myocardial Infarction. Int J Mol Sci. Jan 7 2021;22(2)
12. Vargas KG et al. The functions of estrogen receptor beta in the female brain: A systematic review. Maturitas. Nov 2016;93:41-57.
13. Guida M et al. Cimicifuga racemosa isopropanolic extract for menopausal symptoms: an observational prospective case-control study. Gynecological endocrinology. Dec 2021;37(12):1132-1137.
14. Castelo-Branco C et al. Black cohosh efficacy and safety for menopausal symptoms. The Spanish Menopause Society statement. Gynecological endocrinology. May 2022;38(5):379-384.
15. Castelo-Branco C et al. Review & meta-analysis: isopropanolic black cohosh extract iCR for menopausal symptoms - an update on the evidence. Climacteric. Apr 2021;24(2):109-119.
16. Fernandes ES et al. Effectiveness of the short-term use of Cimicifuga racemosa in the endothelial function of postmenopausal women: a double-blind, randomized, controlled trial. Climacteric. Jun 2020;23(3):245-251.
17. Gorach NV. Effects of cimicifuga racemosa on the hemodynamics parameters and quality of life in perimenopausal women with arterial hypertension. Wiadomosci lekarskie (Warsaw, Poland: 1960). 2018;71(5):1010-1014.
18. Mohapatra S et al. Benefits of Black Cohosh (Cimicifuga racemosa) for Women Health: An Up-Close and In-Depth Review. Pharmaceuticals (Basel). Feb 23 2022;15(3)
19. Pkhaladze L et al. Is More Effective in Combination with Rhodiola rosea L. for Relief of Menopausal Symptoms: A Randomized, Double-Blind, Placebo-Controlled Study. Pharmaceuticals (Basel). May 21 2020;13(5)
20. Rattanatantikul T et al. Efficacy and Safety of Nutraceutical on Menopausal Symptoms in Post-Menopausal Women: A Randomized, Double-Blind, Placebo-Controlled Clinical Trial. J Diet Suppl. 2022;19(2):168-183.
21. Shayan A et al. Effect of Combined Herbal Capsule Menohelp on Hot Flashes and Night Sweats in Postmenopausal Women: A Single-Blind Randomized Controlled Trial. Journal of menopausal medicine. Aug 2020;26(2):135-142.
22. Rotem C et al. Phyto-Female Complex for the relief of hot flushes, night sweats and quality of sleep: randomized, controlled, double-blind pilot study. Gynecological endocrinology. Feb 2007;23(2):117-22.
23. Henneicke-von Zepelin HH. 60 years of Cimicifuga racemosa medicinal products : Clinical research milestones, current study findings and current development. Wien Med Wochenschr. May 2017;167(7-8):147-159.
24. Ruan X et al. Benefit-risk profile of black cohosh (isopropanolic Cimicifuga racemosa extract) with and without St John's wort in breast cancer patients. Climacteric. Aug 2019;22(4):339-347.
25. Chang JL et al. Rheum rhaponticum Extract (ERr 731): Postmarketing Data on Safety Surveillance and Consumer Complaints. Integr Med (Encinitas). Jun 2016;15(3):34-9.
26. Wober J et al. Activation of estrogen receptor-beta by a special extract of Rheum rhaponticum (ERr 731), its aglycones and structurally related compounds. J Steroid Biochem Mol Biol. Nov-Dec 2007;107(3-5):191-201.
27. Moller F et al. Subtype-specific activation of estrogen receptors by a special extract of Rheum rhaponticum (ERr 731), its aglycones and structurally related compounds in U2OS human osteosarcoma cells. Phytomedicine. Nov 2007;14(11):716-26.
28. Kaszkin-Bettag M et al. Confirmation of the efficacy of ERr 731 in perimenopausal women with menopausal symptoms. Altern Ther Health Med. Jan-Feb 2009;15(1):24-34.
29. Heger M et al. Efficacy and safety of a special extract of Rheum rhaponticum (ERr 731) in perimenopausal women with climacteric complaints: a 12-week randomized, double-blind, placebo-controlled trial. Menopause. Sep-Oct 2006;13(5):744-59.
30. Kaszkin-Bettag M et al. The special extract ERr 731 of the roots of Rheum rhaponticum decreases anxiety and improves health state and general well-being in perimenopausal women. Menopause. Mar-Apr 2007;14(2):270-83.
31. Hasper I et al. Long-term efficacy and safety of the special extract ERr 731 of Rheum rhaponticum in perimenopausal women with menopausal symptoms. Menopause. Jan-Feb 2009;16(1):117-31.
32. Shah J et al. Evaluation of the Efficacy and Safety of Rheum rhaponticum Root Extract (ERr 731) for Menopausal Symptoms in Perimenopausal Indian Women: An Interim Analysis. Journal of mid-life health. Apr-Jun 2021;12(2):108-115.
33. Kaszkin-Bettag M et al. Efficacy of the special extract ERr 731 from rhapontic rhubarb for menopausal complaints: a 6-month open observational study. Altern Ther Health Med. Nov-Dec 2008;14(6):32-8.
34. Chen LR et al. Utilization of Isoflavones in Soybeans for Women with Menopausal Syndrome: An Overview. Int J Mol Sci. Mar 22 2021;22(6)
35. Daily JW et al. Equol Decreases Hot Flashes in Postmenopausal Women: A Systematic Review and Meta-Analysis of Randomized Clinical Trials. J Med Food. Feb 2019;22(2):127-139.
36. Crisafulli A et al. Effects of genistein on hot flushes in early postmenopausal women: a randomized, double-blind EPT- and placebo-controlled study. Menopause. Jul-Aug 2004;11(4):400-4.
37. D'Anna R et al. Effects of the phytoestrogen genistein on hot flushes, endometrium, and vaginal epithelium in postmenopausal women: a 2-year randomized, double-blind, placebo-controlled study. Menopause. Mar-Apr 2009;16(2):301-6.
38. Fang K et al. Soy isoflavones and glucose metabolism in menopausal women: A systematic review and meta-analysis of randomized controlled trials. Mol Nutr Food Res. Jul 2016;60(7):1602-14.
39. Baranska A et al. Effects of Soy Protein Containing of Isoflavones and Isoflavones Extract on Plasma Lipid Profile in Postmenopausal Women as a Potential Prevention Factor in Cardiovascular Diseases: Systematic Review and Meta-Analysis of Randomized Controlled Trials. Nutrients. Jul 24 2021;13(8)
40. Sathyapalan T et al. Soy isoflavones improve cardiovascular disease risk markers in women during the early menopause. Nutr Metab Cardiovasc Dis. Jul 2018;28(7):691-697.
41. Boutas I et al. Soy Isoflavones and Breast Cancer Risk: A Meta-analysis. In Vivo. Mar-Apr 2022;36(2):556-562.
42. Touillaud M et al. Use of dietary supplements containing soy isoflavones and breast cancer risk among women aged >50 y: a prospective study. Am J Clin Nutr. Mar 1 2019;109(3):597-605.
43. Leonard LM et al. Maximizing the Estrogenic Potential of Soy Isoflavones through the Gut Microbiome: Implication for Cardiometabolic Health in Postmenopausal Women. Nutrients. Jan 27 2022;14(3)
44. Mayo B et al. Equol: A Bacterial Metabolite from The Daidzein Isoflavone and Its Presumed Beneficial Health Effects. Nutrients. Sep 16 2019;11(9)
45. Sekikawa A et al. Effect of S-equol and Soy Isoflavones on Heart and Brain. Curr Cardiol Rev. 2019;15(2):114-135.
46. Mohsen A et al. Pharmacological and therapeutic properties of the Red Clover (Trifolium pratense L.): an overview of the new finding. J Tradit Chin Med. Aug 2021;41(4):642-649.
47. Kanadys W et al. Evaluation of Clinical Meaningfulness of Red Clover (Trifolium pratense L.) Extract to Relieve Hot Flushes and Menopausal Symptoms in Peri- and Post-Menopausal Women: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Nutrients. Apr 11 2021;13(4)
48. Myers SP et al. Effects of a standardised extract of Trifolium pratense (Promensil) at a dosage of 80mg in the treatment of menopausal hot flushes: A systematic review and meta-analysis. Phytomedicine. Jan 15 2017;24:141-147.
49. Blaszczuk A et al. Role of Phytoestrogen-Rich Bioactive Substances (Linum usitatissimum L., Glycine max L., Trifolium pratense L.) in Cardiovascular Disease Prevention in Postmenopausal Women: A Systematic Review and Meta-Analysis. Nutrients. Jun 14 2022;14(12)
50. Kanadys W et al. Effects of red clover (Trifolium pratense) isoflavones on the lipid profile of perimenopausal and postmenopausal women-A systematic review and meta-analysis. Maturitas. Feb 2020;132:7-16.
51. Wickham KA et al. Short-Term Supplementation With Fermented Red Clover Extract Reduces Vascular Inflammation in Early Post-menopausal Women. Front Cardiovasc Med. 2022;9:826959.
52. Bolton JL et al. The Multiple Biological Targets of Hops and Bioactive Compounds. Chemical research in toxicology. Feb 18 2019;32(2):222-233.
53. Stulikova K et al. Therapeutic Perspectives of 8-Prenylnaringenin, a Potent Phytoestrogen from Hops. Molecules. Mar 15 2018;23(3)
54. van Hunsel F et al Post-Menopausal Vaginal Hemorrhage Related to the Use of a Hop-Containing Phytotherapeutic Product. Drug Saf Case Rep. Dec 2015;2(1):14.
55. Keiler AM et al. Hop extracts and hop substances in treatment of menopausal complaints. Planta Med. May 2013;79(7):576-9.
56. Aghamiri V et al. The effect of Hop (Humulus lupulus L.) on early menopausal symptoms and hot flashes: A randomized placebo-controlled trial. Complement Ther Clin Pract. May 2016;23:130-5.
57. Heyerick A et al. A first prospective, randomized, double-blind, placebo-controlled study on the use of a standardized hop extract to alleviate menopausal discomforts. Maturitas. May 20 2006;54(2):164-75.
58. Erkkola R et al. A randomized, double-blind, placebo-controlled, cross-over pilot study on the use of a standardized hop extract to alleviate menopausal discomforts. Phytomedicine. May 2010;17(6):389-96.
59. Kim HI et al. Efficacy and Safety of a Standardized Soy and Hop Extract on Menopausal Symptoms: A 12-Week, Multicenter, Randomized, Double-Blind, Placebo-Controlled Clinical Trial. J Altern Complement Med. Nov 2021;27(11):959-967.
60. Morali G et al. Open, non-controlled clinical studies to assess the efficacy and safety of a medical device in form of gel topically and intravaginally used in postmenopausal women with genital atrophy. Arzneimittelforschung. 2006;56(3):230-8.
61. Sun W et al. Fenugreek Cultivation with Emphasis on Historical Aspects and its uses in Traditional Medicine and Modern Pharmaceutical Science. Mini Rev Med Chem. 2021;21(6):724-730.
62. Chen Y et al. Advances in the pharmacological activities and mechanisms of diosgenin. Chinese journal of natural medicines. Aug 2015;13(8):578-87.
63. Semwal P et al. Diosgenin: An Updated Pharmacological Review and Therapeutic Perspectives. Oxid Med Cell Longev. 2022;2022:1035441.
64. Khanna A et al. Efficacy of a novel extract of fenugreek seeds in alleviating vasomotor symptoms and depression in perimenopausal women: A randomized, double-blinded, placebo-controlled study. J Food Biochem. Dec 2020;44(12):e13507.
65. Shamshad Begum S et al. A Novel Extract of Fenugreek Husk (FenuSMART) Alleviates Postmenopausal Symptoms and Helps to Establish the Hormonal Balance: A Randomized, Double-Blind, Placebo-Controlled Study. Phytother Res. Nov 2016;30(11):1775-1784.
66. Steels E et al. Efficacy of a Proprietary Trigonella foenum-graecum L. De-Husked Seed Extract in Reducing Menopausal Symptoms in Otherwise Healthy Women: A Double-Blind, Randomized, Placebo-Controlled Study. Phytother Res. Sep 2017;31(9):1316-1322.
67. Mazalzadeh F et al. Effect of Trigonella foenum (fenugreek) vaginal cream on vaginal atrophy in postmenopausal women. J Family Med Prim Care. Jun 2020;9(6):2714-2719.
68. Safary M et al. Comparison of the Effects of Fenugreek Vaginal Cream and Ultra Low- Dose Estrogen on Atrophic Vaginitis. Curr Drug Deliv. 2020;17(9):815-822.
69. Wahab S et al. Glycyrrhiza glabra (Licorice): A Comprehensive Review on Its Phytochemistry, Biological Activities, Clinical Evidence and Toxicology. Plants (Basel). Dec 14 2021;10(12)
70. Minnetti M et al. Effects of licorice on sex hormones and the reproductive system. Nutrition. Nov-Dec 2022;103-104:111727.
71. Markina YV et al. Atheroprotective Effects of Glycyrrhiza glabra L. Molecules. Jul 22 2022;27(15)
72. Azizsoltani A et al. Ethyl Acetate Extract of Licorice Root (Glycyrrhiza glabra) Enhances Proliferation and Osteogenic Differentiation of Human Bone Marrow Mesenchymal Stem Cells. Iran J Pharm Res. Summer 2018;17(3):1057-1067.
73. Boonmuen N et al. Licorice root components in dietary supplements are selective estrogen receptor modulators with a spectrum of estrogenic and anti-estrogenic activities. Steroids. Jan 2016;105:42-9.
74. Hajirahimkhan A et al. Evaluation of estrogenic activity of licorice species in comparison with hops used in botanicals for menopausal symptoms. PLoS One. 2013;8(7):e67947.
75. Mersereau JE et al. Liquiritigenin is a plant-derived highly selective estrogen receptor beta agonist. Mol Cell Endocrinol. Feb 13 2008;283(1-2):49-57.
76. Nahidi F et al. Effects of licorice on relief and recurrence of menopausal hot flashes. Iran J Pharm Res. Spring 2012;11(2):541-8.
77. Menati L et al. Evaluation of contextual and demographic factors on licorice effects on reducing hot flashes in postmenopause women. Health Care Women Int. Jan 2014;35(1):87-99.
78. Sadeghi M et al. Impact of Glycyrrhiza glabra (licorice) vaginal cream on vaginal signs and symptoms of vaginal atrophy in postmenopausal women: A randomized double blind controlled trial. Journal of traditional and complementary medicine. Mar 2020;10(2):110-115.
79. Kwon YJ et al. A Review of the Pharmacological Efficacy and Safety of Licorice Root from Corroborative Clinical Trial Findings. J Med Food. Jan 2020;23(1):12-20.
80. McHugh J et al. A life-threatening case of pseudo-aldosteronism secondary to excessive liquorice ingestion. BMC endocrine disorders. Aug 6 2021;21(1):158.
81. Deutch MR et al. Bioactive Candy: Effects of Licorice on the Cardiovascular System. Foods. Oct 14 2019;8(10)
82. Lin HQ et al. Danggui Buxue Tang (Astragali Radix and Angelicae Sinensis Radix) for menopausal symptoms: A review. J Ethnopharmacol. Mar 6 2017;199:205-210.
83. Hook IL. Danggui to Angelica sinensis root: are potential benefits to European women lost in translation? A review. J Ethnopharmacol. Feb 27 2014;152(1):1-13.
84. Zhang WL et al. Chemical and biological assessment of Angelica herbal decoction: comparison of different preparations during historical applications. Phytomedicine. Aug 15 2012;19(11):1042-8.
85. Circosta C et al. Estrogenic activity of standardized extract of Angelica sinensis. Phytother Res. Aug 2006;20(8):665-9.
86. Haines CJ et al. A randomized, double-blind, placebo-controlled study of the effect of a Chinese herbal medicine preparation (Dang Gui Buxue Tang) on menopausal symptoms in Hong Kong Chinese women. Climacteric. Jun 2008;11(3):244-51.
87. Kupfersztain C et al. The immediate effect of natural plant extract, Angelica sinensis and Matricaria chamomilla (Climex) for the treatment of hot flushes during menopause. A preliminary report. Clinical and experimental obstetrics & gynecology. 2003;30(4):203-6.
88. Hirata JD et al. Does dong quai have estrogenic effects in postmenopausal women? A double-blind, placebo-controlled trial. Fertility and sterility. Dec 1997;68(6):981-6.
89. Yang HM et al. A randomised, double-blind, placebo-controlled trial on the effect of Pycnogenol on the climacteric syndrome in peri-menopausal women. Acta obstetricia et gynecologica Scandinavica. 2007;86(8):978-85.
90. Errichi S et al. Supplementation with Pycnogenol(R) improves signs and symptoms of menopausal transition. Panminerva Med. Sep 2011;53(3 Suppl 1):65-70.
91. Kohama T et al. Effect of low-dose French maritime pine bark extract on climacteric syndrome in 170 perimenopausal women: a randomized, double-blind, placebo-controlled trial. J Reprod Med. Jan-Feb 2013;58(1-2):39-46.
92. Luzzi R et al. Normalization of cardiovascular risk factors in peri-menopausal women with Pycnogenol(R). Minerva ginecologica. Feb 2017;69(1):29-34.
93. Majidi Z et al. Oligopin(R) Supplementation Mitigates Oxidative Stress in Postmenopausal Women with Osteopenia: A Randomized, Double-blind, Placebo-Controlled Trial. Phytomedicine. Jan 2021;81:153417.
94. Cesarone MR et al. Prevention of vaginal dryness in perimenopausal women. Supplementation with Lady Prelox(R). Minerva ginecologica. Dec 2019;71(6):434-441.
95. Marini A et al. Pycnogenol(R) effects on skin elasticity and hydration coincide with increased gene expressions of collagen type I and hyaluronic acid synthase in women. Skin Pharmacol Physiol. 2012;25(2):86-92.
96. Rani A et al. The genus Vitex: A review. Pharmacognosy reviews. Jul 2013;7(14):188-98.
97. Daniele C et al. Vitex agnus castus: a systematic review of adverse events. Drug safety. 2005;28(4):319-32.
98. Chopin Lucks B. Vitex agnus castus essential oil and menopausal balance: a research update [Complementary Therapies in Nursing and Midwifery 8 (2003) 148-154]. Complement Ther Nurs Midwifery. Aug 2003;9(3):157-60.
99. Lucks BC et al. Vitexagnus-castus essential oil and menopausal balance: a self-care survey. Complement Ther Nurs Midwifery. Aug 2002;8(3):148-54.
100. van Die MD et al. Vitex agnus-castus extracts for female reproductive disorders: a systematic review of clinical trials. Planta Med. May 2013;79(7):562-75.
101. Dericks-Tan JS et al. Dose-dependent stimulation of melatonin secretion after administration of Agnus castus. Exp Clin Endocrinol Diabetes. Feb 2003;111(1):44-6.
102. Naseri R et al. Comparison of Vitex agnus-castus Extracts with Placebo in Reducing Menopausal Symptoms: A Randomized Double-Blind Study. Korean J Fam Med. Nov 2019;40(6):362-367.
103. Maffei S et al. Long-term effects of a combination of isoflavones, agnus castus and magnolia extracts on climacteric symptoms and cardiometabolic risk profile in postmenopausal women. Gynecological endocrinology. Apr 2022;38(4):339-344.
104. De Franciscis P et al. Adding Agnus Castus and Magnolia to Soy Isoflavones Relieves Sleep Disturbances Besides Postmenopausal Vasomotor Symptoms-Long Term Safety and Effectiveness. Nutrients. Feb 13 2017;9(2)
105. Molaie M et al. Effects of a combination of Nigella sativa and Vitex agnus-castus with citalopram on healthy menopausal women with hot flashes: results from a subpopulation analysis. Gynecological endocrinology. Jan 2019;35(1):58-61.
106. Peterson B, Nguyen H. St. John's Wort. StatPearls. Publishing LLC.2023.
107. Eatemadnia A et al. The effect of Hypericum perforatum on postmenopausal symptoms and depression: A randomized controlled trial. Complement Ther Med. Aug 2019;45:109-113.
108. Abdali K et al. Effect of St John's wort on severity, frequency, and duration of hot flashes in premenopausal, perimenopausal and postmenopausal women: a randomized, double-blind, placebo-controlled study. Menopause. Mar 2010;17(2):326-31.
109. Liu YR et al. Hypericum perforatum L. preparations for menopause: a meta-analysis of efficacy and safety. Climacteric. Aug 2014;17(4):325-35.
110. Al-Akoum M et al. Effects of Hypericum perforatum (St. John's wort) on hot flashes and quality of life in perimenopausal women: a randomized pilot trial. Menopause. Mar-Apr 2009;16(2):307-14.
111. van Die MD et al. Hypericum perforatum with Vitex agnus-castus in menopausal symptoms: a randomized, controlled trial. Menopause. Jan-Feb 2009;16(1):156-63.
112. van Die MD et al. Effects of a combination of Hypericum perforatum and Vitex agnus-castus on PMS-like symptoms in late-perimenopausal women: findings from a subpopulation analysis. J Altern Complement Med. Sep 2009;15(9):1045-8.
113. Briese V et al. Black cohosh with or without St. John's wort for symptom-specific climacteric treatment--results of a large-scale, controlled, observational study. Maturitas. Aug 20 2007;57(4):405-14.
114. Mahboubi M. Evening Primrose (Oenothera biennis) Oil in Management of Female Ailments. Journal of menopausal medicine. Aug 2019;25(2):74-82.
115. Safdari F et al. Effect of Evening Primrose Oil on Postmenopausal Psychological Symptoms: A Triple-Blind Randomized Clinical Trial. Journal of menopausal medicine. Aug 2021;27(2):58-65.
116. Sharif SN et al. Impact of evening primrose oil consumption on psychological symptoms of postmenopausal women: a randomized double-blinded placebo-controlled clinical trial. Menopause. Feb 2020;27(2):194-198.
117. Farzaneh F et al. The effect of oral evening primrose oil on menopausal hot flashes: a randomized clinical trial. Archives of gynecology and obstetrics. Nov 2013;288(5):1075-9.
118. Kazemi F et al. The Effect of Evening Primrose Oil Capsule on Hot Flashes and Night Sweats in Postmenopausal Women: A Single-Blind Randomized Controlled Trial. Journal of menopausal medicine. Apr 2021;27(1):8-14.
119. Chenoy R et al. Effect of oral gamolenic acid from evening primrose oil on menopausal flushing. BMJ. Feb 19 1994;308(6927):501-3.
120. Jenabi E et al. The effect of Valerian on the severity and frequency of hot flashes: A triple-blind randomized clinical trial. Women Health. Mar 2018;58(3):297-304.
121. Mirabi P et al. The effects of valerian root on hot flashes in menopausal women. Iran J Pharm Res. Winter 2013;12(1):217-22.
122. Taavoni S et al. Effect of valerian on sleep quality in postmenopausal women: a randomized placebo-controlled clinical trial. Menopause. Sep 2011;18(9):951-5.
123. Lee MS et al. Maca (Lepidium meyenii) for treatment of menopausal symptoms: A systematic review. Maturitas. Nov 2011;70(3):227-33.
124. Meissner HO et al. Use of gelatinized maca (lepidium peruvianum) in early postmenopausal women. International journal of biomedical science. Jun 2005;1(1):33-45.
125. Meissner HO et al. Therapeutic Effects of Pre-Gelatinized Maca (Lepidium Peruvianum Chacon) used as a Non-Hormonal Alternative to HRT in Perimenopausal Women - Clinical Pilot Study. International journal of biomedical science. Jun 2006;2(2):143-59.
126. Meissner HO et al. Hormone-Balancing Effect of Pre-Gelatinized Organic Maca (Lepidium peruvianum Chacon): (II) Physiological and Symptomatic Responses of Early-Postmenopausal Women to Standardized doses of Maca in Double Blind, Randomized, Placebo-Controlled, Multi-Centre Clinical Study. International journal of biomedical science. Dec 2006;2(4):360-74.
127. Meissner HO et al. Hormone-Balancing Effect of Pre-Gelatinized Organic Maca (Lepidium peruvianum Chacon): (III) Clinical responses of early-postmenopausal women to Maca in double blind, randomized, Placebo-controlled, crossover configuration, outpatient study. International journal of biomedical science. Dec 2006;2(4):375-94.
128. Brooks NA et al. Beneficial effects of Lepidium meyenii (Maca) on psychological symptoms and measures of sexual dysfunction in postmenopausal women are not related to estrogen or androgen content. Menopause. Nov-Dec 2008;15(6):1157-62.
129. Stojanovska L et al. Maca reduces blood pressure and depression, in a pilot study in postmenopausal women. Climacteric. Feb 2015;18(1):69-78.
130. Ahmad S et al. New Insights into the Biological and Pharmaceutical Properties of Royal Jelly. Int J Mol Sci. Jan 8 2020;21(2)
131. Pasupuleti VR et al. Honey, Propolis, and Royal Jelly: A Comprehensive Review of Their Biological Actions and Health Benefits. Oxid Med Cell Longev. 2017;2017:1259510.
132. Balan A et al. Royal Jelly-A Traditional and Natural Remedy for Postmenopausal Symptoms and Aging-Related Pathologies. Molecules. Jul 20 2020;25(14)
133. Sharif SN et al. Effect of royal jelly on menopausal symptoms: A randomized placebo-controlled clinical trial. Complement Ther Clin Pract. Nov 2019;37:47-50.
134. Asama T et al. Royal Jelly Supplementation Improves Menopausal Symptoms Such as Backache, Low Back Pain, and Anxiety in Postmenopausal Japanese Women. Evid Based Complement Alternat Med. 2018;2018:4868412.
135. Seyyedi F et al. Comparison of the Effects of Vaginal Royal Jelly and Vaginal Estrogen on Quality of Life, Sexual and Urinary Function in Postmenopausal Women. J Clin Diagn Res. May 2016;10(5):QC01-5.
136. Tober C et al. Modulation of neurological pathways by Salvia officinalis and its dependence on manufacturing process and plant parts used. BMC Complement Altern Med. Jun 13 2019;19(1):128.
137. Margetts G et al. Evidence for anti-inflammatory effects and modulation of neurotransmitter metabolism by Salvia officinalis L. BMC Complement Med Ther. May 12 2022;22(1):131.
138. Elgayed SH et al. Estrogenic Effect of Salvia officinalis Extract on Reproductive Function of Female Mice and Identification of Its Phenolic Content. Combinatorial chemistry & high throughput screening. 2021;24(10):1654-1663.
139. Koubaa-Ghorbel F et al. Salvia officinalis mitigates uterus and liver damages induced by an estrogen deficiency in ovariectomized rats. J Food Biochem. May 2021;45(5):e13542.
140. Sabry MM et al. Estrogenic activity of Sage (Salvia officinalis L.) aerial parts and its isolated ferulic acid in immature ovariectomized female rats. J Ethnopharmacol. Jan 10 2022;282:114579.
141. Ghorbani A et al. Pharmacological properties of Salvia officinalis and its components. Journal of traditional and complementary medicine. Oct 2017;7(4):433-440.
142. Dadfar F et al. The effect of Saliva officinalis extract on the menopausal symptoms in postmenopausal women: An RCT. Int J Reprod Biomed. Apr 2019;17(4):287-92.
143. Bommer S et al. First time proof of sage's tolerability and efficacy in menopausal women with hot flushes. Adv Ther. Jun 2011;28(6):490-500.
参考来源:
美国妇女健康办公室
http://www.womenshealth.gov
美国梅奥诊所
www.mayoclinic.org
美国更年期学会
http://www.menopause.org
加拿大卫生部
http://www.hc-sc.gc.ca
加拿大女性健康网
http://www.womenshealthmatters.ca
其他参考文献:详见具体的综合干预方案
免责声明和安全信息
- 本信息(包括任何附带资料)不是为了取代医生或有关合格从业人士的建议或忠告。
- 任何人如果想要对本文涉及的药物、饮食、运动或其他生活方式的使用、或改变调整,以预防或治疗某一特定健康状况或疾病,应首先咨询医生或有关合格从业人士,并获得他/她们的许可。妊娠和哺乳妇女在使用本网站任何内容前,尤其应征求医生的意见。
- 除非另有说明,本网站所述内容仅适用于成人。
- 本网站所推荐的任何产品,消费者应该以实际的产品标签内容为准,尤其应关注重要的安全信息以及产品最新信息,包括剂量、使用方法和禁忌症等。
- 由于循证医学研究、文献及有关产品处于不断的变化中,本网站工作人员将尽力更新。
- 本网站不能保证所载文章内容、综合干预方案以及相关成分或产品述及的健康益处,也不承担任何责任。

